Would K Form A Negative Ion
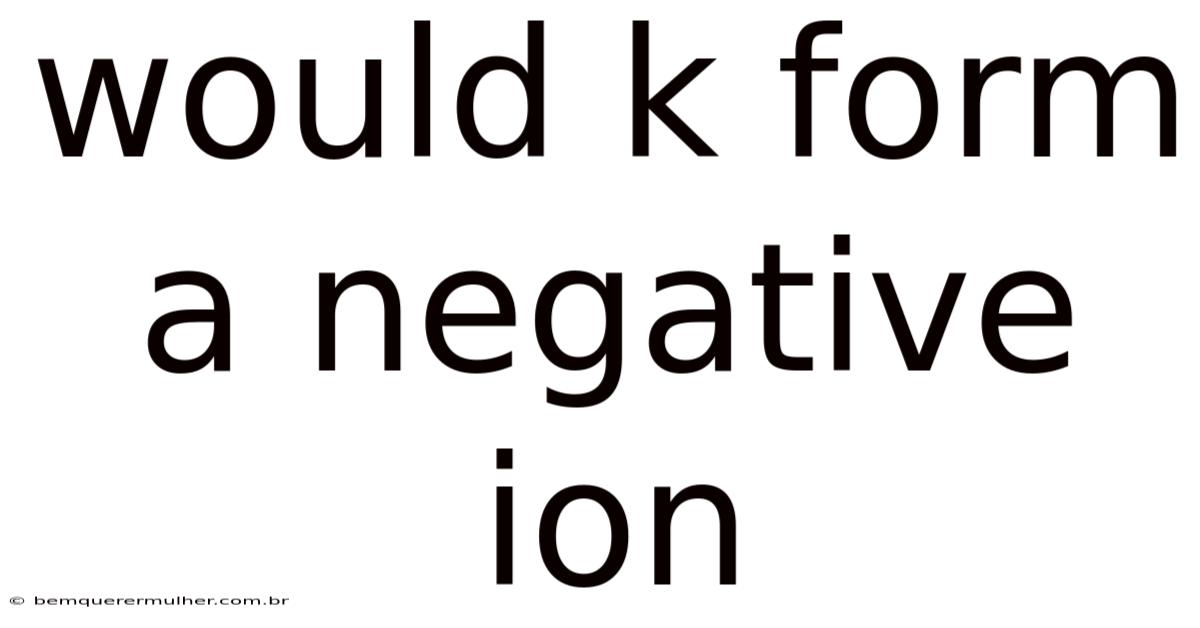
Potassium (K) doesnot form a negative ion. Instead, it consistently forms a positive ion, specifically the potassium ion (K⁺). This fundamental behavior stems directly from the element's position in the periodic table and its inherent chemical properties.
Introduction
Understanding why certain elements form positive ions while others form negative ones is crucial for grasping the basics of chemistry, including ionic bonding, compound formation, and electrical conductivity. Potassium, a highly reactive alkali metal found in Group 1 of the periodic table, exemplifies the formation of a positive ion. This article delves into the reasons behind potassium's preference for forming K⁺ ions, contrasting it with elements that form negative ions, and exploring the consequences of this behavior in compounds and reactions.
Why Potassium Forms Positive Ions (K⁺)
- The Octet Rule and Electron Configuration: Potassium's atomic number is 19. Its electron configuration is [Ar] 4s¹. This means its outermost shell (the 4s orbital) contains only one electron. According to the octet rule, atoms are most stable when their outermost electron shell is filled with 8 electrons (or 2 for the first shell). Potassium achieves this stability by losing this single 4s electron, resulting in a configuration identical to the noble gas argon (Ar) [Ne] 3s² 3p⁶. This loss gives it a full outer shell (now 3s² 3p⁶), satisfying the octet rule and achieving maximum stability.
- Low Ionization Energy: Removing that single 4s electron requires relatively little energy. This property is known as low ionization energy. Potassium has one of the lowest ionization energies among all elements. The ease with which it loses its valence electron makes the formation of K⁺ ions energetically favorable.
- High Electron Affinity: Electron affinity measures an atom's tendency to gain an electron. Potassium has a very low (almost negligible) electron affinity. This means it has little tendency to attract and hold onto an additional electron. In contrast, elements like chlorine (Group 17) have a very high electron affinity, making them eager to gain an electron to form Cl⁻ ions. Potassium's low electron affinity reinforces its preference to lose an electron rather than gain one.
- Electronegativity: Electronegativity is a measure of an atom's ability to attract electrons within a bond. Potassium has a very low electronegativity (0.8 on the Pauling scale). This low value indicates it has little ability to pull electrons towards itself. When forming bonds, especially ionic bonds, potassium readily donates its single valence electron to more electronegative elements, becoming positively charged.
Comparison with Elements Forming Negative Ions
The formation of negative ions (anions) is characteristic of elements that have a high number of valence electrons and a strong desire to achieve a full octet by gaining electrons. Key examples include:
- Halogens (Group 17 - F, Cl, Br, I): These elements have 7 valence electrons. Gaining one electron provides them with a full octet (8 valence electrons), achieving noble gas stability. Their high electron affinity makes them excellent candidates for forming F⁻, Cl⁻, Br⁻, and I⁻ ions.
- Oxygen (Group 16 - O): Oxygen has 6 valence electrons. It commonly gains two electrons to achieve a full octet, forming O²⁻ ions (as in peroxides) or O²⁻ ions (as in oxides). Its high electron affinity supports this behavior.
- Nitrogen (Group 15 - N): Nitrogen has 5 valence electrons. It can gain 3 electrons to form N³⁻ ions (though less common than other anions), or share electrons to form covalent bonds.
The stark contrast lies in their electron configurations and the resulting stability. Potassium (1 valence electron) loses it easily. Halogens (7 valence electrons) gain one electron easily. Oxygen (6 valence electrons) gains two electrons easily. Elements forming negative ions have a high need for electrons to complete their octet, while potassium has a surplus of one electron it readily sheds.
Examples in Compounds and Reactions
The formation of K⁺ ions is fundamental to countless potassium compounds:
- Potassium Chloride (KCl): A classic ionic compound. Potassium metal (K) reacts vigorously with chlorine gas (Cl₂) to form KCl. Potassium loses one electron to form K⁺, while chlorine gains that electron to form Cl⁻. The resulting electrostatic attraction holds the compound together.
- Potassium Hydroxide (KOH): Potassium metal reacts with water (H₂O) to form potassium hydroxide and hydrogen gas. Potassium loses an electron to form K⁺, which then bonds with hydroxide ions (OH⁻) formed when water molecules dissociate.
- Potassium Nitrate (KNO₃): A common fertilizer and explosive component. Potassium nitrate consists of K⁺ ions and NO₃⁻ (nitrate) ions. Potassium again provides the K⁺ cation.
- Biological Systems: Potassium ions (K⁺) are essential electrolytes in the human body, crucial for nerve impulse transmission, muscle contraction, and maintaining fluid balance. Cells actively pump K⁺ in and out to maintain concentration gradients.
Scientific Explanation: The Driving Force
The driving force behind potassium forming K⁺ ions is the pursuit of a stable electron configuration. The energy released when potassium loses its 4s electron and achieves the stable electron configuration of argon (Ar) is significantly greater than the energy required to remove that electron (the ionization energy). This energy difference makes the formation of K⁺ energetically favorable. The resulting K⁺ ion is isoelectronic with argon, possessing a full outer shell of 8 electrons (3s² 3p⁶), which is a highly stable configuration.
FAQ
- Can potassium ever form a negative ion (K⁻)?
- Answer: No, under normal chemical conditions, potassium does not form a K⁻ ion. Its low ionization energy and high electron affinity make losing an electron far more favorable than gaining one. The K⁻ ion would have a configuration of [Ar] 4s² 4p⁶, which is less stable than the noble gas configuration of Ar ([Ne] 3s² 3p⁶). While highly theoretical or under extreme conditions, K⁻ is not observed in stable compounds or reactions.
- Why does potassium form positive ions while chlorine forms negative ions?
- Answer: Potassium (Group 1) has 1 valence electron and a strong drive to lose it for stability, resulting in K⁺. Chlorine (Group 17) has 7 valence electrons and a strong drive to gain one electron for stability, resulting in Cl⁻. Their
Their contrasting electronegativities drive this complementary behavior. Potassium’s low electronegativity (≈0.82 on the Pauling scale) means it readily relinquishes its lone 4s electron, whereas chlorine’s high electronegativity (≈3.16) makes it eager to accept an electron to fill its valence shell. When the two meet, the electron transfer is almost instantaneous, producing the stable ionic lattice of KCl where each K⁺ is surrounded by six Cl⁻ ions and vice‑versa, maximizing electrostatic attraction and minimizing lattice energy.
Additional FAQs
-
How does hydration affect K⁺ in aqueous solutions?
When K⁺ dissolves in water, it becomes surrounded by a hydration shell of water molecules oriented with their oxygen atoms toward the cation. This solvation stabilizes the ion in solution, reduces its effective charge, and facilitates its mobility, which is essential for its role in biological conductivity and electrochemical gradients. -
Are there any notable exceptions where potassium exhibits oxidation states other than +1?
Under extreme conditions—such as in matrix‑isolated species or under high‑pressure synthesis—transient K⁰ or K⁻ species have been spectroscopically detected, but no bulk, isolable compounds exist where potassium deviates from the +1 oxidation state. Its chemistry remains overwhelmingly monovalent due to the large energy penalty associated with removing a second electron (the second ionization energy is >30 eV). -
What safety considerations arise when handling potassium metal?
Potassium reacts violently with water, producing KOH and flammable H₂ gas, and can ignite spontaneously in moist air. It must be stored under inert hydrocarbons (e.g., mineral oil) or in a dry argon atmosphere, and handled with tools that avoid spark generation. Protective equipment, including gloves, goggles, and flame‑resistant lab coats, is mandatory.
Conclusion
The propensity of potassium to form K⁺ ions stems from its electronic structure: a single, loosely held 4s electron whose removal yields a stable, argon‑like configuration. This energetic favorability underpins the formation of ubiquitous ionic compounds such as KCl, KOH, and KNO₃, and sustains vital physiological processes where K⁺ gradients drive nerve impulses and muscle contractions. While potassium’s chemistry is dominated by the +1 oxidation state, understanding its ionization behavior, solvation effects, and reactive hazards provides a comprehensive picture of why this alkali metal is both indispensable in industry and essential to life.
Latest Posts
Latest Posts
-
What Is A Step In Practicing Correct Personal Hygiene
Mar 23, 2026
-
Which Of The Following Is Accurate Concerning Nonverbal Communication
Mar 23, 2026
-
Which Of The Following Does Not Reduce Shrink
Mar 23, 2026
-
Which Area Is Inappropriate For Storing Food
Mar 23, 2026
-
Pagar Means Pain To Owe Pagan To Pay
Mar 23, 2026