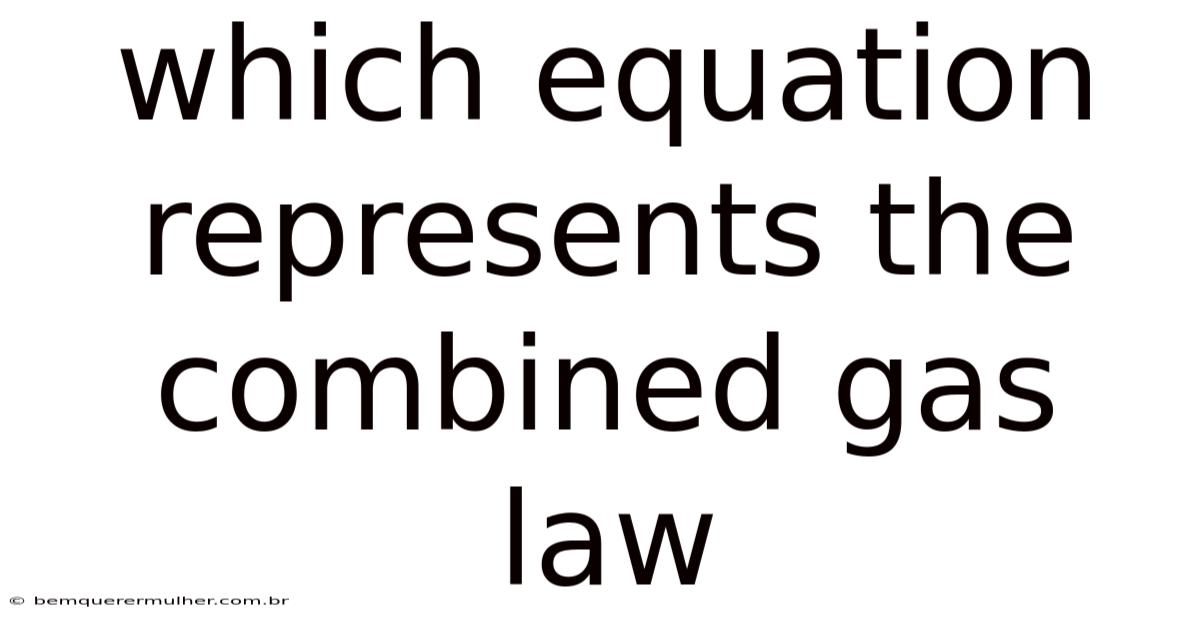The combined gas law integrates the individualgas laws—Boyle’s law, Charles’s law, and Gay‑Lussac’s law—into a single relationship that describes how pressure, volume, and temperature of a fixed amount of gas behave together. Which equation represents the combined gas law? The answer is the expression
[\frac{P_1 V_1}{T_1}= \frac{P_2 V_2}{T_2} ]
where (P) stands for pressure, (V) for volume, and (T) for absolute temperature (in kelvin). This equation tells us that when the amount of gas remains constant, the ratio of pressure to temperature multiplied by volume stays the same before and after a change. Simply put, if any two of the three variables shift, the third must adjust in a predictable way to keep the ratio constant. This principle is foundational for solving problems in chemistry, physics, and engineering that involve gas behavior under varying conditions Easy to understand, harder to ignore..
Introduction to Gas Laws
Before diving into the combined form, it helps to review the three simpler laws that precede it:
- Boyle’s Law – At constant temperature, pressure and volume are inversely proportional ((P_1 V_1 = P_2 V_2)).
- Charles’s Law – At constant pressure, volume and temperature are directly proportional ((V_1/T_1 = V_2/T_2)).
- Gay‑Lussac’s Law – At constant volume, pressure and temperature are directly proportional ((P_1/T_1 = P_2/T_2)).
Each of these relationships captures a specific pair of variables while holding the third steady. Worth adding: the combined gas law simply removes that restriction, allowing all three variables to change simultaneously. Understanding this integration is essential for anyone tackling real‑world scenarios such as scuba diving, weather forecasting, or industrial gas storage.
Deriving the Combined Equation
To see which equation represents the combined gas law in practice, start with the three individual relationships:
- From Boyle’s law: (P_1 V_1 = P_2 V_2) (temperature constant).
- From Charles’s law: (V_1/T_1 = V_2/T_2) (pressure constant).
- From Gay‑Lussac’s law: (P_1/T_1 = P_2/T_2) (volume constant).
By algebraic manipulation—multiplying the three equations together and canceling common terms—you arrive at:
[ \frac{P_1 V_1}{T_1}= \frac{P_2 V_2}{T_2} ]
This derivation illustrates why the combined gas law is sometimes written as:
[ \frac{P_1 V_1}{T_1}=k \quad\text{and}\quad \frac{P_2 V_2}{T_2}=k ]
where (k) is a constant for a given sample of gas. The constancy of (k) underscores the law’s predictive power: if you know any two of the new conditions, you can solve for the third.
Practical Applications
Knowing which equation represents the combined gas law enables scientists and engineers to calculate unknowns in a variety of contexts:
- Scuba diving – When a diver ascends, ambient pressure drops. Using the combined gas law, divers can estimate how their air‑filled tanks will expand or compress, helping them manage decompression schedules safely.
- Weather balloons – As a balloon rises, external pressure falls while the gas inside expands. The combined gas law predicts the balloon’s volume increase until the surrounding air density matches the balloon’s average density, causing it to burst.
- Industrial reactors – In chemical plants, temperature and pressure are often adjusted to optimize reaction rates. Engineers use the combined gas law to forecast how changing conditions will affect gas volume, ensuring that storage tanks are neither over‑pressurized nor under‑utilized.
In each case, the equation serves as a bridge between measurable quantities and the underlying physics of the system Surprisingly effective..
Common Misconceptions
Even though the combined gas law is straightforward, several misunderstandings persist:
-
Misconception 1: “The law works for any gas, regardless of its state.” Reality: The law assumes the gas behaves ideally—meaning molecules have negligible volume and no intermolecular forces. Real gases deviate under high pressure or low temperature, so corrections (e.g., van der Waals equation) may be needed.
-
Misconception 2: “Temperature can be used in Celsius or Fahrenheit directly.”
Reality: The law requires absolute temperature (kelvin). Using Celsius would produce incorrect ratios because the zero point is arbitrary It's one of those things that adds up.. -
Misconception 3: “If pressure doubles, volume must halve, regardless of temperature changes.”
Reality: Volume also depends on temperature. If temperature rises proportionally, the volume change may be less dramatic The details matter here..
Recognizing these pitfalls helps students apply the law correctly and avoid costly errors in laboratory or fieldwork.
Frequently Asked Questions
Q1: What units should be used for pressure, volume, and temperature?
A: Pressure can be expressed in atmospheres (atm), pascals (Pa), or torr; volume in liters (L) or cubic meters (m³); temperature must be in kelvin (K). Consistency across the initial and final states is essential Simple, but easy to overlook. Took long enough..
Q2: Can the combined gas law be applied to mixtures of gases? A: Yes, provided the mixture behaves ideally and the total amount of gas (in moles) remains unchanged. Each component’s partial pressure contributes to the overall pressure in the equation.
Q3: How does the combined gas law differ from the ideal gas law?
A: The ideal gas law ((PV = nRT)) includes the amount of gas (n) and the universal gas constant (R). The combined gas law is a simplified version that assumes a constant amount of gas, focusing only on the relationship among (P), (V), and (T) And that's really what it comes down to..
Q4: Is the combined gas law useful for solids or liquids?
A: No. The law specifically addresses gases where volume changes significantly with pressure and temperature. Solids and liquids are relatively incompressible, so their volume changes are negligible under typical conditions That's the part that actually makes a difference. No workaround needed..
Conclusion
The quest to discover which equation represents the combined gas law leads directly to the elegant relationship (\frac{P_1 V_1}{T_1}= \frac{P_2 V_2}{T_2}). This formula condenses three fundamental gas laws into a single, versatile tool that scientists, engineers, and students use to predict how gases respond when pressure, volume, and temperature shift together. By mastering its derivation, applications, and the assumptions behind it
Not the most exciting part, but easily the most useful Practical, not theoretical..
, one gains a deeper appreciation for the interplay of physical variables that govern the behavior of gases—a cornerstone of chemistry, physics, and engineering disciplines. The combined gas law, while simple in form, encapsulates the essence of gas behavior under varying conditions, serving as an indispensable resource in both academic study and practical applications. Whether in the design of chemical processes, the operation of engines, or the analysis of atmospheric changes, this law remains a testament to the power of scientific principles in understanding and manipulating the natural world.
The combined gas law is more than a textbook formula; it is a practical bridge that lets scientists and engineers translate a change in one variable into predictable changes in the others. By treating pressure, volume, and temperature as interdependent, it provides a quick, reliable check for experimental data, a diagnostic tool for troubleshooting equipment, and a foundational element for more advanced models that incorporate non‑ideal behavior That's the part that actually makes a difference..
In practice, the law’s simplicity is its greatest asset. A single equation can be applied to a sealed syringe, a scuba tank, a weather balloon, or a chemical reactor—any system where the amount of gas stays fixed and the gas behaves close to ideal. When the conditions push the gas beyond the realm of ideality, the combined gas law still offers a baseline from which corrections can be applied, whether through virial expansions, real‑gas equations of state, or empirical adjustments.
The bottom line: mastering the combined gas law equips one with a versatile lens for viewing the dynamic world of gases. It reminds us that even the most complex systems obey clear, quantitative relationships, and that a deep understanding of these relationships empowers us to predict, control, and innovate across science and engineering.
End of Article