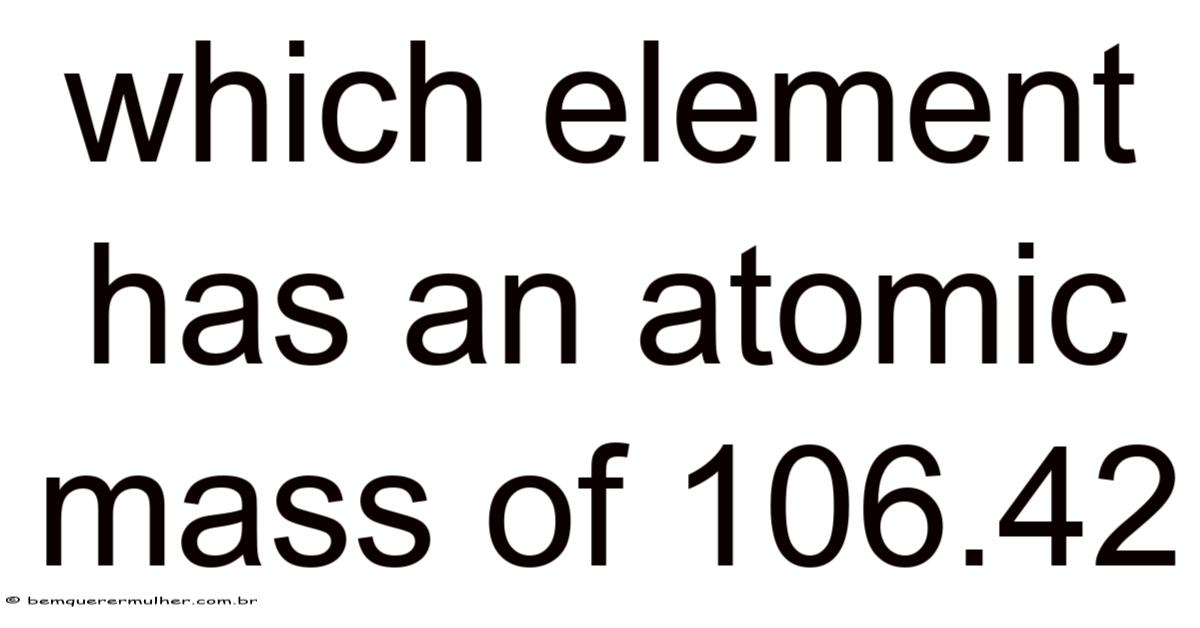Which Element Has an Atomic Mass of 106.42?
The atomic mass of an element is a fundamental property that defines its identity on the periodic table. When we refer to the atomic mass of 106.That said, 42, we are pointing to a specific synthetic element that resides in the sixth period of the periodic table. This element, with its unique atomic weight, plays a significant role in the study of synthetic chemistry and nuclear physics.
Understanding Atomic Mass and Its Significance
Atomic mass, also known as relative atomic mass, is the average mass of an element’s isotopes, weighted by their natural abundance. And it is measured in atomic mass units (u) and is typically expressed as a decimal value. In real terms, for example, the atomic mass of iron is approximately 55. Plus, 85 u, reflecting the average of its naturally occurring isotopes. On top of that, in the case of the element with an atomic mass of 106. 42, this value represents the calculated average of its isotopes, primarily derived from synthetic production methods Most people skip this — try not to..
The Element with Atomic Mass 106.42: Seaborgium (Sg)
The element with an atomic mass of 106.Day to day, 42 is seaborgium, denoted by the symbol Sg. Seaborgium is a synthetic element with the atomic number 106, meaning it has 106 protons in its nucleus. It was discovered in 1974 by a team of scientists at the University of California, Berkeley, led by Glenn T. Seaborg, a renowned chemist who later won the Nobel Prize in Chemistry. The element was named in honor of Seaborg’s contributions to the discovery of multiple synthetic elements, including americium, curium, and californium.
Key Properties of Seaborgium
Seaborgium is a transuranium element, belonging to the actinide series. It is a superheavy element, which means it has an atomic number greater than 103. Here are some of its notable properties:
- Electron Configuration: [Rn] 5f¹⁴ 6d⁴ 7s² 7p²
- State at Room Temperature: Predicted to be a solid
- Melting Point: Estimated to be around 700°C
- Boiling Point: Approximately 1,400°C
Seaborgium is part of the 6d transition series, and its chemistry is still largely theoretical due to its scarcity and short half-life. It is produced artificially in particle accelerators by bombarding heavy elements like lead or bismuth with high-energy ions.
Discovery and Naming of Seaborgium
The discovery of seaborgium was a collaborative effort between scientists at Berkeley and the Joint Institute for Nuclear Research in Dubna, Russia. The element was first synthesized through nuclear reactions involving the bombardment of californium-249 with copper ions. The resulting compound, californium-249 + copper-63, produced seaborgium-252, which decayed within milliseconds.
Short version: it depends. Long version — keep reading.
The name "seaborgium" was chosen to recognize Glenn Seaborg’s pioneering work in the synthesis of transuranium elements. The symbol Sg was selected to represent the element, following the convention of using the discoverer’s name for newly created elements.
Why Is Seaborgium Important?
Seaborgium holds significance in the study of nuclear chemistry and the exploration of the island of stability, a theoretical region of superheavy elements that may have longer half-lives. Researchers investigate seaborgium and similar elements to understand the limits of nuclear stability and the behavior of matter under extreme conditions That's the whole idea..
This is the bit that actually matters in practice The details matter here..
Additionally, seaborgium’s atomic mass of 106.42 is critical in calculations involving nuclear reactions and isotopic analysis. While it has no stable isotopes, its properties provide insights into the periodic trends and the behavior of elements in the actinide series.
Frequently Asked Questions (FAQ)
What is the difference between atomic mass and mass number?
The mass number is the total number of protons and neutrons in an atom’s nucleus, while atomic mass is the weighted average of all isotopes of an element, considering their natural abundance. For seaborgium, the mass number varies depending on the isotope, but its atomic mass is standardized at 106.42.
Is seaborgium found in nature?
No, seaborgium is a synthetic element and does not occur naturally. It is produced artificially in laboratories through nuclear reactions and exists for only fractions of a second before decaying No workaround needed..
What are the health effects of seaborgium?
Due to its extremely short half-life and radioactivity, seaborgium poses no practical health risks. It is not encountered in everyday life and is studied only in controlled research environments Practical, not theoretical..
How does seaborgium fit into the periodic table?
Seaborgium is located in group 6 of the periodic table, below molybdenum, tungsten, and chromium. It is part of the actinide series, which includes elements with atomic numbers greater than 89. Its placement reflects its electron configuration and chemical properties.
Conclusion
The element with an atomic mass of 106.42 is seaborgium, a synthetic superheavy element that represents a remarkable achievement in nuclear chemistry. Its discovery and study have expanded our understanding of the periodic table and the behavior of matter under extreme conditions. While seaborgium exists only in trace amounts and for brief periods, its atomic mass and properties continue to inspire scientific inquiry into the frontiers of chemistry and physics And that's really what it comes down to..