Number Of Moles 4.01g Of Ch4
bemquerermulher
Mar 13, 2026 · 6 min read
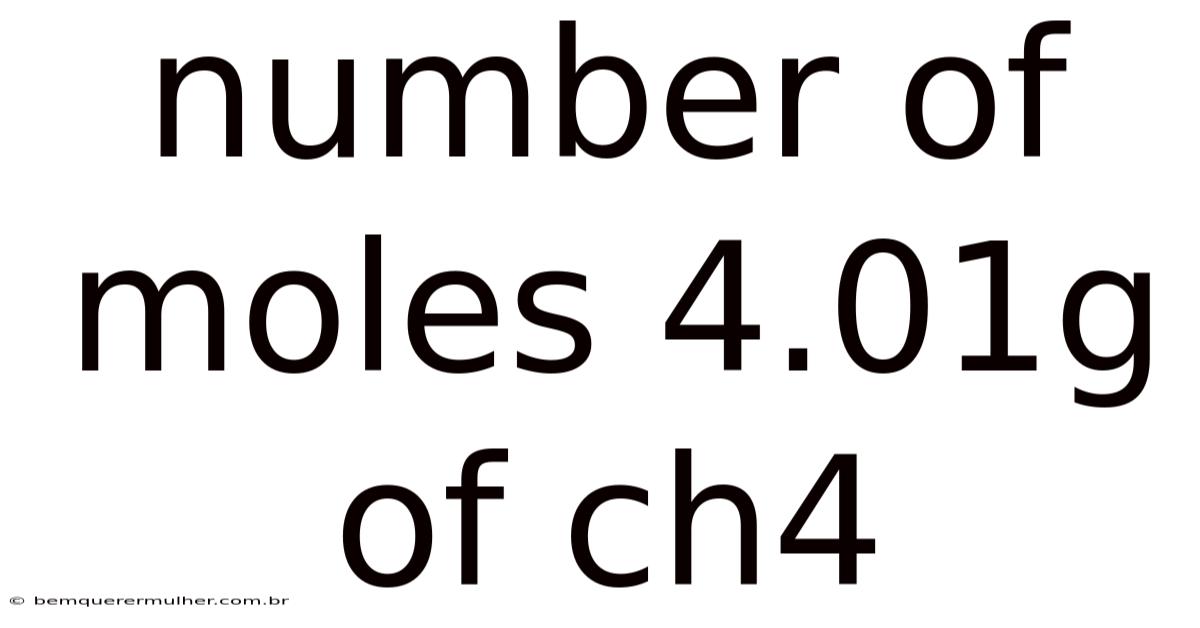
Table of Contents
Calculating Moles: A Step-by-Step Guide to 4.01g of CH₄
Understanding the concept of the mole is fundamental to mastering chemistry. It is the bridge between the microscopic world of atoms and molecules and the measurable, macroscopic world we work in the laboratory. This article will demystify this crucial concept by performing a complete, detailed calculation: determining the exact number of moles in a 4.01-gram sample of methane (CH₄). By the end, you will not only know the answer for this specific case but will possess a transferable skill essential for any chemistry student or enthusiast.
What is a Mole? The Chemist's "Dozen"
Before diving into calculations, we must define our unit. A mole (symbol: mol) is the SI base unit for amount of substance. One mole of any substance contains exactly 6.022 x 10²³ elementary entities (atoms, molecules, ions, etc.). This number is known as Avogadro's constant (Nₐ).
Think of it this way: a "dozen" means 12 items. A "mole" means 6.022 x 10²³ items. The power of the mole lies in its direct connection to mass. The mass of one mole of a substance (its molar mass) in grams is numerically equal to the average mass of one molecule or formula unit in atomic mass units (amu). For carbon, the atomic mass is 12.01 amu, so one mole of carbon atoms has a mass of 12.01 grams. This elegant relationship is the key to all mole calculations.
Step 1: Determine the Molar Mass of Methane (CH₄)
Our first task is to calculate the molar mass of methane. Methane is a covalent molecule consisting of one carbon atom and four hydrogen atoms. We obtain the atomic masses from the periodic table.
- Atomic mass of Carbon (C): 12.01 g/mol
- Atomic mass of Hydrogen (H): 1.008 g/mol
The formula for methane is CH₄. Therefore, its molar mass (M) is calculated as: M(CH₄) = (1 × atomic mass of C) + (4 × atomic mass of H) M(CH₄) = (1 × 12.01 g/mol) + (4 × 1.008 g/mol) M(CH₄) = 12.01 g/mol + 4.032 g/mol M(CH₄) = 16.042 g/mol
We will use this value, 16.042 g/mol, as the conversion factor. It tells us that every single mole of methane molecules weighs exactly 16.042 grams.
Step 2: The Core Calculation – Using the Conversion Formula
The relationship between mass (m), molar mass (M), and number of moles (n) is defined by the simple but powerful formula:
n = m / M
Where:
- n = number of moles (mol)
- m = given mass of the substance (g)
- M = molar mass of the substance (g/mol)
This formula is the cornerstone of quantitative chemistry. It states that the number of moles is equal to the mass you have divided by the mass of one mole of that substance.
Now, we plug in our specific values:
- m = 4.01 g (the mass of our methane sample)
- M = 16.042 g/mol (the molar mass we calculated)
n = 4.01 g / 16.042 g/mol
Performing the division: n ≈ 0.2500 mol
The precise calculation yields: 4.01 / 16.042 = 0.2500 (to four significant figures).
Step 3: Interpreting the Result and Connecting to Avogadro's Number
Our calculation shows that 4.01 grams of methane is exactly 0.2500 moles.
What does this mean on a molecular level? Since one mole contains 6.022 x 10²³ molecules, 0.2500 moles contains: Number of CH₄ molecules = 0.2500 mol × (6.022 x 10²³ molecules/mol) Number of CH₄ molecules = 1.5055 x 10²³ molecules
Therefore, your 4.01-gram sample of methane gas contains approximately 150.55 sextillion individual methane molecules. This illustrates the staggering scale of Avogadro's number—a mass we can easily handle on a bench scale corresponds to an almost incomprehensibly large number of molecules.
The Significance of the Numbers: Why 4.01g?
The choice of 4.01g is not arbitrary; it is pedagogically brilliant. Notice the molar mass of methane is 16.042 g/mol. If you take one-quarter (¼) of that molar mass: 16.042 g/mol ÷ 4 = 4.0105 g
Our given mass, 4.01g, is essentially one-quarter of the molar mass of methane (rounded to three significant figures). Therefore, 4.01 g of CH₄ is exactly 0.250 mol, or one-quarter of a mole. This creates a perfect, memorable example: if the molar mass is X grams per mole, then X/4 grams is 0.25 moles. This pattern helps students build intuition. For any substance, if you have a mass equal to one-fourth of its molar mass, you have a quarter-mole.
Common Mistakes and Pitfalls to Avoid
Even with a straightforward example, errors can occur. Here are the most frequent pitfalls:
- Forgetting to Calculate Molar Mass: The most critical step is determining the correct molar mass from the chemical formula. Never assume the molar mass is the same as another compound. For CH₄, you must account for four hydrogen atoms.
- Incorrect Atomic Mass Values: Always use the most recent values from your periodic table (typically with two decimal places, e.g., C=12.01, H=1.008). Using whole numbers (C=12, H=1) will give an approximate answer (4.00g / 16.00g/mol = 0.250 mol) but is not precise enough for formal work.
- Unit Mismatch: The formula
n = m/Monly works if mass is in grams and molar mass is in g/mol. If your mass is given in milligrams
(mg) or kilograms, convert to grams first.
-
Significant Figure Errors: The mass (4.01 g) has three significant figures, and the molar mass (16.042 g/mol) has five. The quotient should be reported with three significant figures: 0.250 mol. Reporting 0.2500 mol (four figures) overstates the precision implied by the given mass.
-
Confusing Moles with Mass: Remember, a mole is a count (like a dozen), not a mass. Saying "0.250 moles of methane" is correct; saying "0.250 moles of grams" is nonsensical.
Conclusion: The Mole as a Fundamental Bridge
This exercise, from calculating the molar mass of methane to determining the number of molecules in a 4.01-gram sample, demonstrates the profound utility of the mole concept. It is the essential bridge connecting the tangible, measurable world of grams and liters to the invisible realm of atoms and molecules. The deliberate choice of a quarter-mole mass provides a clear, memorable anchor for students, reinforcing that the molar mass in grams defines the mass of one mole of any substance. Mastering this simple calculation—accurately finding molar mass, applying the formula n = m/M, and respecting units and significant figures—is not an academic triviality. It is the first and most critical step toward quantitative chemistry. Every subsequent topic, from stoichiometry and solution concentration to reaction yields and gas laws, rests upon this foundational ability to convert seamlessly between mass and amount. Ultimately, understanding the mole empowers us to weigh out precise quantities of matter, predict the outcomes of chemical reactions, and comprehend the material world in terms of its fundamental particulate nature.
Latest Posts
Latest Posts
-
If Wxyz Is A Square Which Statements Must Be True
Mar 13, 2026
-
Actions To Take When Capture Is Imminent Include
Mar 13, 2026
-
A Burn Can Be Which Of The Following Colors
Mar 13, 2026
-
Formed To Draft Formal Petitions Of Protest To Parliament
Mar 13, 2026
-
Investing In Mid Cap And Large Cap Companies Means
Mar 13, 2026
Related Post
Thank you for visiting our website which covers about Number Of Moles 4.01g Of Ch4 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.