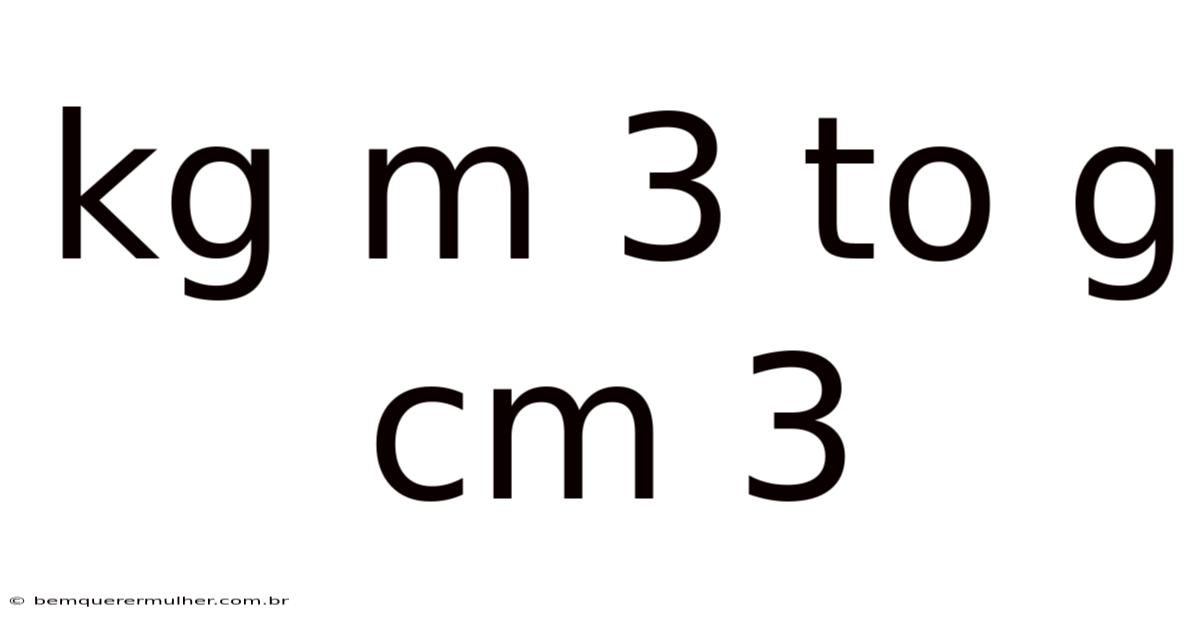Converting kg/m³ to g/cm³: A Complete Guide to Density Unit Conversion
Understanding how to convert between kilograms per cubic meter (kg/m³) and grams per cubic centimeter (g/cm³) is a fundamental skill in science, engineering, and everyday applications. Think about it: 001 g/cm³**. In practice, mastering this simple yet critical conversion ensures accuracy in calculations from material science to fluid dynamics. The core relationship is straightforward: **1 kg/m³ is exactly equal to 0.So naturally, this conversion is essential whenever you work with density, the measure of mass per unit volume. Worth adding: while both units describe the same physical property, they operate on vastly different scales. This article will demystify the math, provide clear step-by-step methods, explore the scientific reasoning behind the factor, and offer practical examples to solidify your understanding, ensuring you can confidently switch between these units whenever necessary Small thing, real impact..
Understanding Density Units: kg/m³ and g/cm³
What is Density?
At its core, density (ρ) is defined as mass divided by volume. The formula is ρ = m/V. It tells us how much "stuff" is packed into a given space. A material with high density, like lead, has a lot of mass in a small volume. A material with low density, like foam, has little mass in the same volume. This concept is central in identifying substances, calculating buoyancy, designing structures, and understanding atmospheric phenomena Simple, but easy to overlook..
The Units: kg/m³ and g/cm³
The two most common units for density in the metric system are:
- Kilograms per cubic meter (kg/m³): This is the SI (International System of Units) derived unit for density. It is standard in scientific literature, engineering specifications, and most global technical contexts. One kg/m³ means one kilogram of mass occupies a volume of one cubic meter.
- Grams per cubic centimeter (g/cm³): This unit is exceptionally common in chemistry, material science, and everyday use in many countries. It is often more intuitive for smaller, laboratory-scale samples. One g/cm³ means one gram of mass occupies a volume of one cubic centimeter.
The key difference lies in the scale of the mass unit (kilogram vs. gram) and the volume unit (cubic meter vs. cubic centimeter). A kilogram is 1000 times heavier than a gram. Here's the thing — a cubic meter is a much larger volume than a cubic centimeter—in fact, 1 m³ = 1,000,000 cm³. The conversion factor accounts for both of these differences simultaneously.
The Conversion Factor Explained: Why 0.001?
Breaking Down the Math
The conversion isn't arbitrary; it's a direct result of the definitions of the base units. Let's derive it logically:
- Mass Conversion: 1 kilogram (kg) = 1000 grams (g).
- Volume Conversion: 1 meter (m) = 100 centimeters (cm). Because of this, 1 cubic meter (m³) = (100 cm) * (100 cm) * (100 cm) = 1,000,000 cubic centimeters (cm³).
Now, we combine these for density:
- Start with 1 kg/m³. Which means * Convert the mass: 1 kg = 1000 g. Day to day, * Convert the volume: 1 m³ = 1,000,000 cm³. * So, 1 kg/m³ = (1000 g) / (1,000,000 cm³).
Simplify the fraction: 1000 / 1,000,000 = 1 / 1000 = 0.001 Worth knowing..
Which means, 1 kg/m³ = 0.001 g/cm³.
Conversely, to go from g/cm³ to kg/m³, you multiply by 1000. Even so, for example, the density of water is approximately 1 g/cm³. To express this in SI units: 1 g/cm³ * 1000 = 1000 kg/m³ That's the part that actually makes a difference. Which is the point..
Why This Matters: The Scale Difference
This factor of 1000 highlights a crucial point: g/cm³ is a much larger unit than kg/m³. A density of 1 g/cm³ sounds small and manageable for a lab sample. Its equivalent, 1000 kg/m³, sounds larger and is more appropriate for describing bulk materials like concrete or soil. Using the wrong unit can lead to orders-of-magnitude errors in engineering calculations or scientific experiments. Recognizing that a value in g/cm³ will be 1000 times smaller numerically than its equivalent in kg/m³ is a vital sanity check.
Step-by-Step Conversion Guide
Method 1: Direct
When transitioning between these units, it's helpful to follow a systematic approach. Let’s say you have a density value in g/cm³ and want to convert it to kg/m³. First, understand the relationship between the two dimensions: mass and volume. Worth adding: since density (ρ) is defined as mass per unit volume, the conversion hinges on expressing both measurements in compatible units. Take this case: if you have a substance with a density of 2 g/cm³, you can convert it directly by multiplying by 1000 to shift the scale to kg/m³. This is because 1 g/cm³ = 1000 kg/m³. This direct method ensures consistency across all calculations.
If you prefer a more manual process, start by identifying the numerical value in g/cm³. As an example, 0.5 g/cm³ becomes 0.Which means divide it by 1000 to move it into the kg/m³ range. 5 ÷ 1000 = 0.Worth adding: 0005 kg/m³, which is equivalent to 500 kg/m³. This method reinforces the understanding of the underlying unit transformation.
Practical Applications
In real-world scenarios, such conversions are essential. Suppose a geologist measures the porosity of a rock sample in g/cm³ and needs to express it in kg/m³ for structural analysis. By applying the conversion logic, the result will align with the required framework for engineering or physics. Similarly, in chemistry, when preparing solutions, knowing the density in g/cm³ helps determine the mass needed for a given volume, which is crucial for accurate mixing and concentration.
Understanding these nuances ensures precision in experiments, designs, and data interpretation. The interplay between these units underscores the importance of unit awareness in scientific communication.
So, to summarize, mastering the conversion between density units fosters clarity and accuracy across disciplines. Whether you're adjusting measurements for research or troubleshooting technical specs, these principles remain indispensable.
Conclusion: Grasping the relationships between these density units not only streamlines calculations but also enhances your ability to apply scientific knowledge confidently. Stay vigilant with conversions, and you'll find they are as essential as the principles they support.
Common Pitfalls to Avoid
Despite the straightforward nature of density conversions, several frequent mistakes can compromise accuracy. One of the most prevalent errors involves confusing the direction of conversion—multiplying when division is required, or vice versa. Another common issue stems from neglecting significant figures, particularly when working with precise scientific measurements where rounding errors can propagate through subsequent calculations. Additionally, failing to verify unit consistency before performing calculations often leads to erroneous results. As an example, mixing metric prefixes without proper conversion can introduce systematic errors that are difficult to detect without careful review Still holds up..
Quick Reference Summary
To consolidate the conversion process, remember these key points:
- 1 g/cm³ = 1,000 kg/m³
- To convert g/cm³ to kg/m³: multiply by 1,000
- To convert kg/m³ to g/cm³: divide by 1,000
This relationship applies universally regardless of the material being measured, whether it's water, concrete, metals, or organic compounds.
Final Thoughts
Density unit conversion exemplifies a broader principle in scientific and engineering practice: attention to fundamental details ensures reliable outcomes. The ability to naturally handle between g/cm³ and kg/m³ not only demonstrates technical competence but also prevents costly errors in design, manufacturing, and research. As you continue applying these conversions in your work, they will become second nature—a testament to the power of consistent practice and thorough understanding The details matter here..
Mastering such fundamental conversions is more than a computational skill; it is a cornerstone of scientific rigor that empowers professionals across fields to communicate precisely, design confidently, and innovate effectively.
Practical Applications Across Industries
Understanding density conversions extends far beyond textbook exercises, finding critical application in numerous professional fields. In the construction industry, engineers routinely convert between g/cm³ and kg/m³ when specifying materials for concrete mixtures, asphalt formulations, and structural component design. The automotive and aerospace sectors rely on precise density calculations to optimize weight-to-strength ratios, directly impacting fuel efficiency and performance capabilities.
In the pharmaceutical and food industries, density conversions ensure formulation accuracy, dosage consistency, and quality control. Laboratory technicians working with chemicals, solutions, and biological materials must frequently translate between units to maintain experimental reproducibility and comply with international standards Worth keeping that in mind..
Environmental scientists apply these conversions when analyzing soil composition, water quality, and pollution concentrations. Meanwhile, material scientists and metallurgists depend on accurate density measurements for alloy development and material characterization.
Teaching Strategies for Mastery
Educators seeking to impart these conversion skills effectively often employ multiple pedagogical approaches. Which means visual demonstrations using materials of known density—such as water, aluminum, and gold—help students grasp the physical significance of numerical values. Dimensional analysis worksheets reinforce the mathematical relationships, while real-world problem sets contextualize the learning within authentic scenarios.
Interactive digital tools and smartphone applications have emerged as valuable supplements to traditional instruction, providing instant feedback and allowing learners to explore hypothetical scenarios without time constraints.
Looking Ahead: Digital Transformation in Unit Conversion
The future of scientific measurement and conversion continues evolving with advancing technology. But smart sensors now embedded in industrial equipment can automatically perform unit conversions, reducing human error in manufacturing and research settings. Cloud-based databases maintain standardized conversion factors, ensuring consistency across global collaborations.
Quick note before moving on.
Artificial intelligence systems increasingly assist scientists and engineers by automatically detecting unit mismatches, suggesting appropriate conversions, and flagging potential errors before they propagate through calculations. These technological advances complement rather than replace the fundamental understanding that professionals must maintain.
Whether you find yourself in a research laboratory, manufacturing facility, classroom, or engineering office, the principles of density unit conversion remain a constant companion. By internalizing these relationships and avoiding common pitfalls, you equip yourself with a foundational skill that transcends specific applications—transforming raw data into meaningful insights that drive progress across every scientific endeavor.
The integration of these digital tools is poised to further streamline workflows and enhance accuracy. Imagine a scenario where a researcher designing a new pharmaceutical formulation can instantly convert milligram quantities of a drug ingredient into grams, all within a user-friendly interface. Practically speaking, or consider the impact on environmental monitoring, where real-time data from sensors can be automatically converted to standardized units, facilitating comprehensive analysis and reporting. These advancements represent a significant step towards a more efficient and reliable scientific landscape.
Quick note before moving on.
On the flip side, it's crucial to remember that technology is a tool, not a replacement for understanding. Day to day, critical thinking and the ability to interpret results in context are very important. That's why while AI can automate certain aspects of conversion, the underlying principles of density, volume, and mass must remain firmly grasped. The most effective approach will likely involve a synergistic blend of human expertise and technological assistance, allowing professionals to focus on higher-level analysis and innovation.
This is the bit that actually matters in practice.
All in all, density unit conversion is a fundamental skill with far-reaching implications across numerous scientific disciplines. The evolution of digital tools offers exciting possibilities for enhancing accuracy and efficiency, but the core principles remain essential. By embracing these advancements while maintaining a solid foundation in the underlying mathematics, individuals can confidently handle the complexities of scientific measurement and contribute to meaningful discoveries that shape our world.