How Many Particles Equals 8.1 Mol Of C2h4o
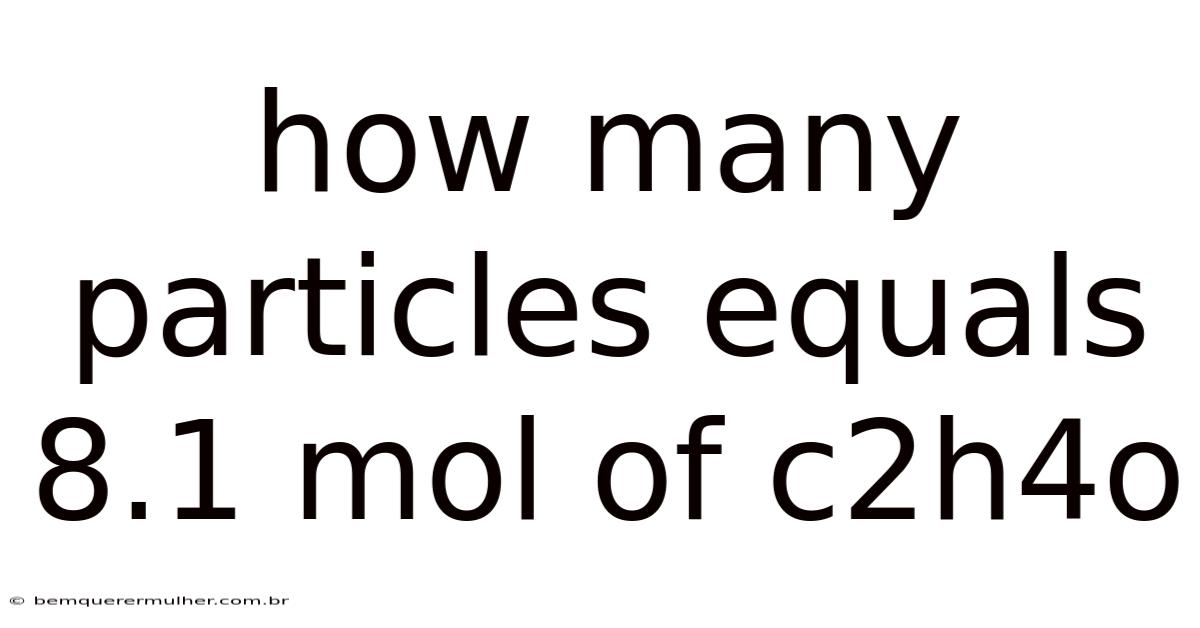
How Many Particles Are in 8.1 Moles of C₂H₄O? A Deep Dive into Avogadro's Number
Understanding the relationship between the macroscopic world we can see and the atomic-scale world we cannot is one of the most powerful concepts in chemistry. At the heart of this connection lies a single, elegant number: Avogadro's constant. When we ask, "How many particles are in 8.1 moles of C₂H₄O?" we are not just performing a calculation; we are bridging that gap between grams and atoms, between laboratory measurements and the fundamental building blocks of matter. This article will unpack this question completely, providing the precise answer while building a robust conceptual framework that applies to any substance.
The Foundation: What is a Mole?
Before calculating, we must solidify the core concept. A mole (abbreviated as mol) is the SI base unit for amount of substance. One mole of any substance contains exactly 6.02214076×10²³ elementary entities. This number is Avogadro's constant (Nₐ), named after the scientist Amedeo Avogadro.
The "elementary entities" can be atoms, molecules, ions, electrons, or any other specified group. For a covalent compound like C₂H₄O, the relevant entity is the molecule. Therefore, the key relationship is:
1 mole of C₂H₄O molecules = 6.02214076×10²³ C₂H₄O molecules
This constant acts as a universal conversion factor between the amount we measure in moles and the actual count of particles.
Decoding the Formula: C₂H₄O
The formula C₂H₄O represents a molecular formula, telling us each molecule of this substance is composed of:
- 2 carbon (C) atoms
- 4 hydrogen (H) atoms
- 1 oxygen (O) atom
It is crucial to recognize that this formula corresponds to more than one possible compound, known as isomers. The most common are:
- Acetaldehyde (CH₃CHO): An aldehyde.
- Ethylene Oxide (C₂H₄O): A cyclic ether.
For the purpose of counting molecules, the isomer does not matter. One molecule of acetaldehyde and one molecule of ethylene oxide are both counted as one particle of C₂H₄O. The calculation of the number of molecules is identical regardless of which specific structure we are considering. The chemical properties differ, but the particle count per mole does not.
The Core Calculation: A Direct Proportionality
The calculation is a straightforward application of the definition of a mole. We set up a proportion:
Number of particles = (Number of moles) × (Avogadro's constant)
Plugging in our values:
- Number of moles = 8.1 mol
- Avogadro's constant = 6.02214076×10²³ mol⁻¹
Therefore: Number of C₂H₄O molecules = 8.1 mol × (6.02214076×10²³ molecules/mol)
Performing the multiplication: 8.1 × 6.02214076×10²³ = 48.779340156×10²³
Expressing this in proper scientific notation (with one non-zero digit before the decimal): 4.8779340156×10²⁴ molecules
The Critical Role of Significant Figures
Our raw calculation yields a long number, but we must consider significant figures. The given value "8.1 mol" has two significant figures. Avogadro's constant is a defined fundamental constant with an infinite number of significant figures for this purpose. Therefore, our final answer must be rounded to two significant figures.
- 4.8779340156×10²⁴ rounded to two significant figures is 4.9×10²⁴.
Final Answer: 8.1 moles of C₂H₄O contains approximately 4.9×10²⁴ molecules.
This means if you had 8.1 moles of acetaldehyde, you would have nearly 5 billion billion billion individual acetaldehyde molecules. It is an astronomically large number, illustrating why the mole is such a practical unit—it allows chemists to work with amounts they can measure in the lab (grams, liters) while knowing exactly how many particles they are dealing with.
Step-by-Step Breakdown for Clarity
Let's isolate the process for any similar problem:
- Identify the particle: Determine what constitutes a single "particle" for the given substance. For C₂H₄O, it is a molecule.
- Recall the conversion factor: 1 mol = 6.022×10²³ particles (using the common rounded value for calculation, but remembering the exact value for precision).
- Multiply: Amount in moles × Avogadro's number.
- Apply significant figures: Round the result to match the least precise measurement in the problem. Here, 8.1 (two sig figs) dictates the answer.
Example with Rounded Constant: 8.1 mol × 6.022×10²³ mol⁻¹ = 48.7782×10²³ = 4.87782×10²⁴ ≈ 4.9×10²⁴ molecules.
Frequently Asked Questions (FAQ)
Q1: Does it matter if C₂H₄O is acetaldehyde or ethylene oxide? No. The mole counts molecules. One molecule of either compound is one particle. The calculation for the number of molecules is identical. The difference would only matter if you were calculating the number of atoms (which would be different per molecule) or the mass.
Q2: What if the question asked for atoms instead of molecules? You would first find the number of molecules (as we did), then multiply by the number of atoms per molecule. For C₂H₄O, each molecule contains 2 + 4 + 1 = 7 atoms. Total atoms = (4.9×10²⁴ molecules) × (7 atoms/molecule) = 3.43×10²⁵ atoms (rounded to two sig figs).
This foundational calculation extends directly into stoichiometry, where mole ratios from balanced equations allow chemists to predict quantities of reactants and products. Knowing that 8.1 moles of a substance corresponds to ~4.9×10²⁴ molecules provides the essential link between measurable mass and the actual particle interactions driving chemical change. It transforms abstract coefficients into tangible counts of collisions and transformations.
Furthermore, this principle underpins concepts like molarity (moles per liter) and empirical formula determination. When analyzing a compound's composition, converting percentage masses to mole ratios—and then to molecule ratios—reveals the simplest whole-number formula. The ability to move seamlessly between moles, molecules, and atoms is thus central to both quantitative analysis and theoretical understanding in chemistry.
In summary, the mole and Avogadro's number serve as the indispensable conversion factor between the human-scale world of grams and liters and the atomic-scale world of particles. Mastering this conversion, with careful attention to significant figures and particle identity, is not merely an exercise but a core competency for any chemist. It empowers precise communication, accurate prediction, and a deeper appreciation of the immense, yet countable, universe within every sample of matter.
Latest Posts
Latest Posts
-
Which Of The Following Applies To The Electron
Mar 22, 2026
-
From The Following Choices Select The Factors
Mar 22, 2026
-
Find The Range Of The Following Piecewise Function
Mar 22, 2026
-
Fill In The Information Missing From This Table
Mar 22, 2026
-
How Many Cups Is 60 Oz
Mar 22, 2026