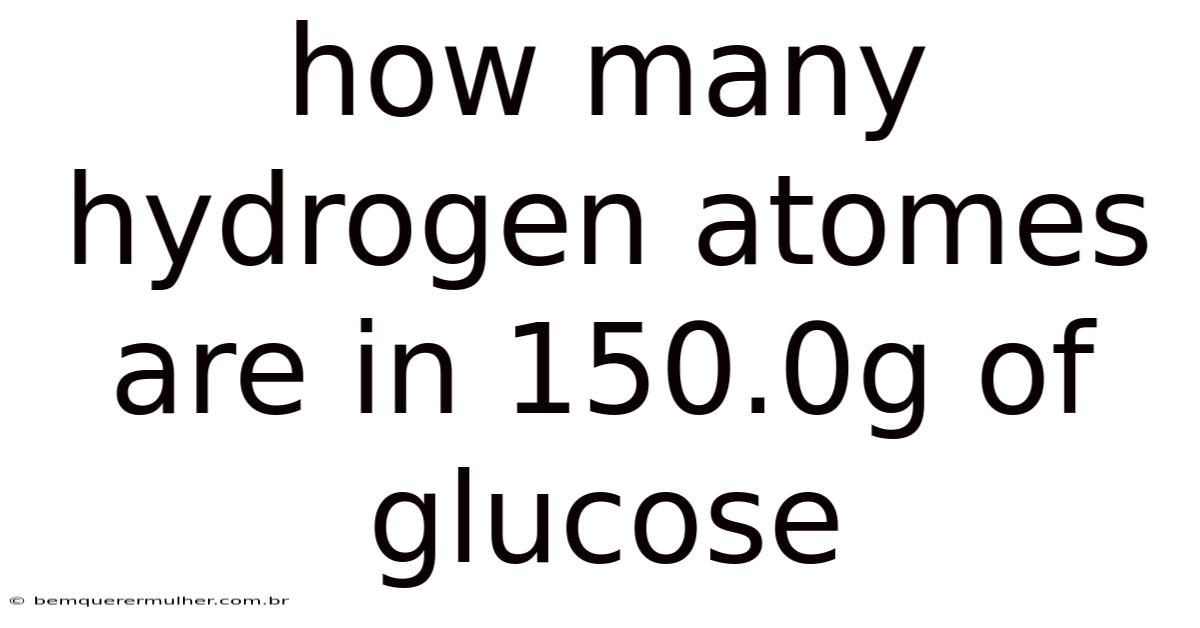How Many HydrogenAtoms Are in 150.0g of Glucose? A Step-by-Step Breakdown
When tackling a question like how many hydrogen atoms are in 150.0g of glucose, it’s essential to approach it systematically. The calculation involves converting mass to moles, determining the number of hydrogen atoms per molecule, and scaling up using Avogadro’s number. Glucose (C₆H₁₂O₆) is a simple sugar found in living organisms, and understanding its molecular composition is key to solving this problem. This process not only answers the question but also reinforces fundamental chemistry principles Practical, not theoretical..
Step 1: Understand the Chemical Formula of Glucose
Glucose has the molecular formula C₆H₁₂O₆, meaning each molecule contains 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms. Consider this: this formula is critical because it tells us that for every molecule of glucose, there are 12 hydrogen atoms. Without this information, the calculation would be impossible Worth keeping that in mind..
The first step in solving the problem is to recognize that the mass of glucose (150.Which means 0g) must be converted into moles. This requires knowing the molar mass of glucose, which is calculated by summing the atomic masses of all atoms in the molecule.
Step 2: Calculate the Molar Mass of Glucose
The molar mass of a compound is the sum of the atomic masses of its constituent elements. Practically speaking, using standard atomic masses:
- Carbon (C): 12. 01 g/mol
- Hydrogen (H): 1.008 g/mol
- Oxygen (O): 16.
For glucose (C₆H₁₂O₆):
- Carbon: 6 × 12.01 = 72.06 g/mol
- Hydrogen: 12 × 1.008 = 12.096 g/mol
- Oxygen: 6 × 16.00 = 96.
Adding these together:
**72.06
- 12.096 + 96.00 = 180.156 g/mol
This value represents the mass of one mole of glucose Not complicated — just consistent..
Step 3: Convert Mass to Moles
To find the number of moles in 150.0g of glucose, divide the given mass by the molar mass:
[ \text{Moles of glucose} = \frac{150.0 , \text{g}}{180.156 , \text{g/mol}} \approx 0 Not complicated — just consistent..
Step 4: Determine the Number of Hydrogen Atoms
Each molecule of glucose contains 12 hydrogen atoms. To find the total number of hydrogen atoms, multiply the number of moles of glucose by Avogadro’s number (6.022 × 10²³ molecules/mol) and then by 12:
[ \text{Number of hydrogen atoms} = 0.8326 , \text{mol} \times 6.022 \times 10^{23} , \text{molecules/mol} \times 12 ]
[ = 0.8326 \times 6.022 \times 10^{23} \times 12 ]
[ = 6.022 \times 10^{23} \times 9.9912 ]
[ \approx 6.01 \times 10^{24} , \text{hydrogen atoms} ]
Conclusion
By following these steps—understanding the molecular formula, calculating the molar mass, converting mass to moles, and scaling up using Avogadro’s number—we determined that 150.Now, 0g of glucose contains approximately 6. 01 × 10²⁴ hydrogen atoms. Plus, this calculation not only answers the question but also demonstrates the practical application of stoichiometry and the mole concept in chemistry. Such problems are foundational for understanding molecular composition and are widely applicable in fields like biochemistry and nutrition.
This analytical process underscores the importance of precision in chemical calculations. Every detail, from molecular structure to atomic weight, plays a important role in deriving meaningful results. By systematically breaking down each component, we not only solve the problem but also reinforce the interconnectedness of scientific principles.
Further, recognizing how these calculations influence real-world applications—whether in medical research or food science—highlights their relevance beyond the laboratory. The final numbers may seem vast, but they represent tangible insights into the behavior of molecules.
Pulling it all together, mastering the formula and methodology behind the glucose calculation empowers us to tackle similar challenges with confidence. Consider this: such exercises strengthen our grasp of chemistry, emphasizing the value of accuracy and logical reasoning. Embracing these lessons ensures we remain adept at interpreting data and applying it effectively Most people skip this — try not to. That alone is useful..
Conclusion: The journey through this calculation reinforces the significance of foundational chemistry concepts and their practical implications.
Conclusion
By following these steps—understanding the molecular formula, calculating the molar mass, converting mass to moles, and scaling up using Avogadro’s number—we determined that 150.This calculation not only answers the question but also demonstrates the practical application of stoichiometry and the mole concept in chemistry. Which means 0g of glucose contains approximately 6. Also, 01 × 10²⁴ hydrogen atoms. Such problems are foundational for understanding molecular composition and are widely applicable in fields like biochemistry and nutrition.
This analytical process underscores the importance of precision in chemical calculations. Every detail, from molecular structure to atomic weight, plays a critical role in deriving meaningful results. By systematically breaking down each component, we not only solve the problem but also reinforce the interconnectedness of scientific principles. Further, recognizing how these calculations influence real-world applications—whether in medical research or food science—highlights their relevance beyond the laboratory. The final numbers may seem vast, but they represent tangible insights into the behavior of molecules.
All in all, mastering the formula and methodology behind the glucose calculation empowers us to tackle similar challenges with confidence. Such exercises strengthen our grasp of chemistry, emphasizing the value of accuracy and logical reasoning. Embracing these lessons ensures we remain adept at interpreting data and applying it effectively. Also, the journey through this calculation reinforces the significance of foundational chemistry concepts and their practical implications. It’s a testament to how seemingly simple molecular structures hold within them a wealth of information, readily accessible through careful application of fundamental chemical principles.
The interplay of theory and application continues to shape scientific progress. Which means as disciplines converge, their synergy reveals deeper truths. Such efforts bridge abstract concepts with tangible outcomes, fostering a shared understanding Easy to understand, harder to ignore..
Pulling it all together, navigating these complexities demands vigilance and adaptability. Even so, the insights gained remain a cornerstone for advancing knowledge across disciplines. The bottom line: such endeavors illuminate the enduring impact of foundational science, bridging past and future advancements That's the whole idea..
Conclusion: Such pursuits solidify the enduring relevance of chemistry in shaping our world.
The ability to quantify atomiccomponents within a substance is not merely an academic exercise; it is a cornerstone of innovation and problem-solving across disciplines. Take this case: in pharmaceuticals, precise calculations of molecular components are critical for drug formulation, ensuring the correct dosage and efficacy of medications. Even so, similarly, in environmental science, understanding the distribution of elements in compounds aids in assessing pollution levels and developing remediation strategies. The calculation of hydrogen atoms in glucose, for example, could extend to analyzing the hydrogen content in biofuels or metabolic byproducts, offering insights into sustainable energy solutions or human health Nothing fancy..
Worth adding, this exercise exemplifies how foundational chemistry principles underpin advancements in technology. Because of that, from nanotechnology to artificial intelligence, the ability to model and predict molecular behavior relies on the same rigorous methodologies. By mastering these calculations, we equip ourselves to address complex challenges, whether in developing new materials, optimizing industrial processes, or advancing biomedical research. The sheer scale of numbers involved—like the vast number of hydrogen atoms in a seemingly simple molecule—reminds us that even the smallest components can have profound implications when viewed through the lens of scientific inquiry.
In the long run, the journey from a molecular formula to a tangible result underscores the power of systematic thinking. It is a testament to how abstract concepts, when applied with care and precision, can yield solutions that impact both scientific understanding and practical outcomes. Think about it: as we continue to explore the complexities of matter, the lessons learned from such calculations remain vital, serving as a bridge between curiosity and application. In a world increasingly driven by data and innovation, the ability to dissect and interpret molecular structures is not just a skill—it is a vital tool for progress.
Conclusion
The calculation of hydrogen atoms in glucose, while seemingly straightforward, encapsulates the essence of chemical reasoning and its far-reaching applications. It highlights the meticulous nature of scientific work, where every step—from molecular analysis to large-scale conversions—contributes to a deeper understanding of the natural world. Beyond the numbers, this process reinforces the importance of critical thinking, adaptability, and the interconnectedness of scientific disciplines. As we refine our methods and expand our knowledge, such exercises remind us that even the most basic questions can lead to transformative insights. In embracing these principles, we not only solve problems but also contribute to a legacy of innovation that shapes the future. The enduring relevance of chemistry lies in its ability to transform complexity into clarity, and through such calculations, we continue to tap into the mysteries of the molecular world.