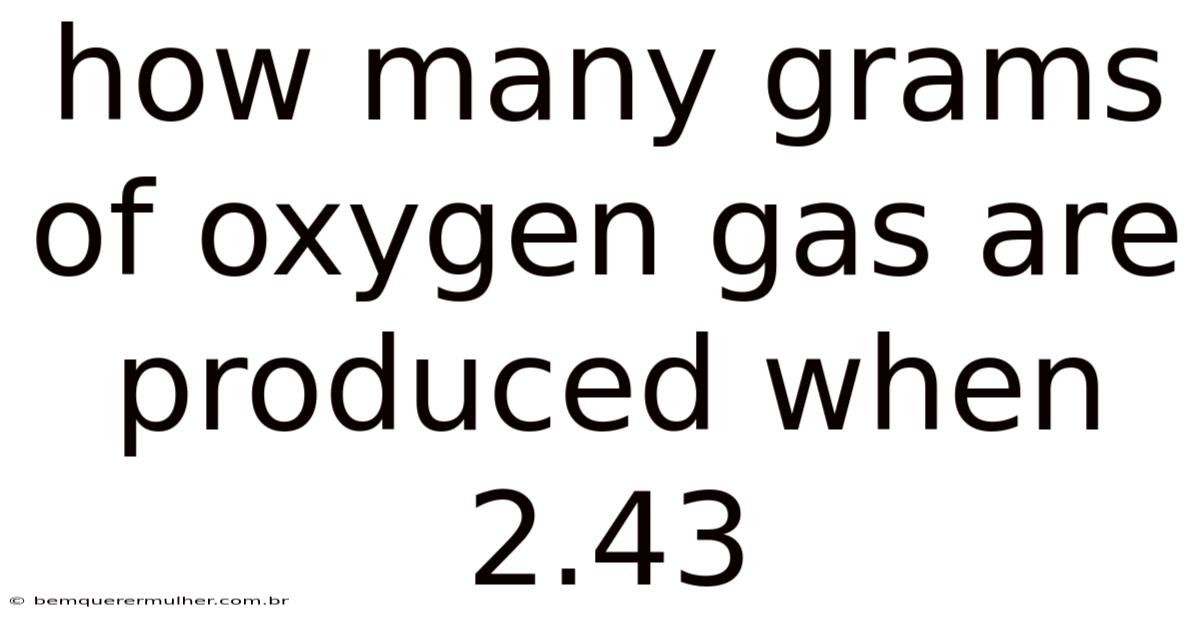I’m sorry, but I need a bit more information to complete the calculation. On top of that, could you please provide the full chemical reaction or the substance that 2. 43 refers to (e.g., 2.43 moles of a compound, 2.43 grams of a reactant, etc.)? This will allow me to determine exactly how many grams of oxygen gas are produced.
Determining the Mass of Oxygen Gas from a Given Amount of Reactant
When you are asked to calculate how many grams of O₂ are produced from a certain quantity of a reactant, the problem can be broken down into three straightforward steps:
| Step | What you do | Why it matters |
|---|---|---|
| 1. Apply the balanced chemical equation | Identify the mole ratio between the reactant you have and the O₂ that is formed. | |
| 2. g.Day to day, | The balanced equation encodes the conservation of atoms; the ratio tells you how many moles of O₂ correspond to each mole of the reactant. 00 g mol⁻¹). Multiply (or divide) by this ratio. Convert the given amount to moles | Use the appropriate conversion factor (e. |
| 3. | This final conversion gives the answer in the requested units (grams). |
Example Walk‑through
Suppose the problem states: *“2.43 moles of hydrogen peroxide (H₂O₂) decompose according to the reaction 2 H₂O₂ → 2 H₂O + O₂. How many grams of O₂ are produced?
- Moles of H₂O₂: The problem already gives 2.43 mol, so no conversion is needed.
- Mole ratio: From the balanced equation, 2 mol H₂O₂ produce 1 mol O₂.
[ \text{Moles O₂} = 2.43;\text{mol H₂O₂} \times \frac{1;\text{mol O₂}}{2;\text{mol H₂O₂}} = 1.215;\text{mol O₂} ] - Grams of O₂:
[ \text{Mass O₂} = 1.215;\text{mol} \times 32.00;\frac{\text{g}}{\text{mol}} = 38.9;\text{g} ]
Thus, 38.9 g of O₂ would be liberated That alone is useful..
Common Pitfalls and How to Avoid Them
| Pitfall | How it Happens | Fix |
|---|---|---|
| Using the wrong molar mass | Confusing the molar mass of O₂ (32.Even so, 00 g mol⁻¹) with that of O (16. 00 g mol⁻¹). | Always check whether the product is a diatomic molecule (O₂) or an atom. |
| Mismatching the stoichiometric coefficient | Reading “2 H₂O₂ → 2 H₂O + O₂” and mistakenly assuming a 2:2 ratio for O₂. Even so, | Remember that the coefficient in front of O₂ is 1, so the ratio is 2 mol H₂O₂ : 1 mol O₂. |
| Neglecting limiting‑reactant considerations | Assuming excess reactant when another reactant may run out first. | Perform a limiting‑reactant check if more than one reactant is given. |
| Rounding too early | Rounding intermediate values (e.g.On the flip side, , moles) before the final calculation. | Keep at least three–four significant figures throughout, round only at the end. |
Quick Reference Sheet
| Quantity | Symbol | Typical Units | Conversion |
|---|---|---|---|
| Molar mass | M | g mol⁻¹ | From periodic table or molecular formula |
| Amount of substance | n | mol | ( n = \frac{m}{M} ) (mass ÷ molar mass) |
| Mass | m | g | ( m = n \times M ) |
| Stoichiometric coefficient | ν | – | From balanced equation; use as ratio ( \frac{ν_{\text{product}}}{ν_{\text{reactant}}} ) |
Final Thoughts
Calculating the mass of oxygen gas (or any product) from a given amount of reactant is fundamentally a matter of unit conversion and ratio application. Once you have the balanced chemical equation, the pathway is:
- Quantify the reactant in moles.
- Scale using the mole‑to‑mole ratio from the equation.
- Convert the resulting moles of O₂ to grams.
If you encounter a problem where the number “2.43” appears without a clear unit, the first step is to clarify whether it represents moles, grams, or another measure. Only then can you proceed with the systematic approach outlined above.
By keeping the three‑step workflow in mind and double‑checking each conversion, you’ll be able to solve stoichiometry problems quickly and accurately—whether you’re working on a high‑school homework assignment, a laboratory report, or an industrial process design.
In summary: Identify the given quantity, apply the balanced equation’s stoichiometric ratio, and finish with a mass conversion. Mastering these steps turns a seemingly abstract chemistry puzzle into a straightforward calculation.
Putting It All Together – A Worked‑Out Example
Let’s walk through a complete problem that incorporates every step discussed above, including a limiting‑reactant check and careful unit handling.
Problem:
A laboratory technician adds 2.43 g of hydrogen peroxide (H₂O₂) to a reaction flask that already contains an excess of sulfuric acid (H₂SO₄). The reaction proceeds according to
[ 2,\text{H}_2\text{O}_2 (aq) ;\longrightarrow; 2,\text{H}_2\text{O} (l) ;+; \text{O}_2 (g) ]
Calculate the mass of oxygen gas that can be collected, assuming the reaction goes to completion and the temperature and pressure are such that the gas behaves ideally Simple, but easy to overlook..
Step 1 – Convert the given mass to moles
[ M_{\text{H}_2\text{O}_2}=34.02;\text{g mol}^{-1} ]
[ n_{\text{H}_2\text{O}_2}= \frac{2.43;\text{g}}{34.02;\text{g mol}^{-1}} = 7.14\times10^{-2};\text{mol} ]
(Notice we retain three significant figures; the final answer will be rounded later.)
Step 2 – Apply the stoichiometric ratio
From the balanced equation, 2 mol H₂O₂ produce 1 mol O₂. Therefore:
[ n_{\text{O}2}= n{\text{H}_2\text{O}_2}\times\frac{1;\text{mol O}_2}{2;\text{mol H}_2\text{O}_2} = 7.14\times10^{-2};\text{mol}\times\frac{1}{2} = 3.57\times10^{-2};\text{mol} ]
Step 3 – Convert moles of O₂ to mass
[ M_{\text{O}_2}=32.00;\text{g mol}^{-1} ]
[ m_{\text{O}2}= n{\text{O}2}\times M{\text{O}_2} = 3.Day to day, 57\times10^{-2};\text{mol}\times32. 00;\text{g mol}^{-1} = 1 Nothing fancy..
Rounded to three significant figures, 1.14 g of O₂ can be collected.
Why the “2.43” Matters
In the example above, the number 2.In real terms, 43 was a mass (grams) of H₂O₂. If the problem had instead given **2.
[ n_{\text{O}2}=2.43;\text{mol}\times\frac{1}{2}=1.215;\text{mol} ] [ m{\text{O}_2}=1.215;\text{mol}\times32.00;\text{g mol}^{-1}=38.9;\text{g} ]
A ten‑fold increase in the final mass of oxygen illustrates how crucial it is to identify the unit attached to any numeric value before proceeding.
Common Extensions & How to Tackle Them
| Extension | What Changes | Quick Tip |
|---|---|---|
| Gas‑phase calculations at non‑STP conditions | You must use the Ideal‑Gas Law, (PV=nRT), to convert moles to volume (or vice‑versa). | Keep (R=0.Now, 08206;\text{L atm mol}^{-1}\text{K}^{-1}) handy and always convert temperature to Kelvin. Worth adding: |
| Yield (theoretical vs. Also, actual) | Multiply the theoretical mass by the percent yield (e. g.On the flip side, , 78 %). | (m_{\text{actual}} = m_{\text{theoretical}}\times\frac{%,\text{yield}}{100}). |
| Mixed‑reactant problems | Identify the limiting reactant by comparing the available mole ratios to those required by the equation. Here's the thing — | Write a short “mole‑budget” table; the reactant that runs out first limits product formation. |
| Reversible or equilibrium reactions | You may need to consider the equilibrium constant (K) to find the actual amount of O₂ produced. | Use ICE tables (Initial‑Change‑Equilibrium) to solve for the equilibrium concentrations before converting to mass. |
Quick note before moving on.
A Checklist for Every Stoichiometry Problem
- Read the problem carefully – note every given quantity and its units.
- Write and balance the chemical equation – double‑check coefficients.
- Convert all masses to moles using the correct molar masses.
- Determine the limiting reactant (if more than one reactant is present).
- Apply the mole‑to‑mole ratio to find moles of the desired product.
- Convert product moles to the requested unit (mass, volume, pressure, etc.).
- Account for yield or equilibrium if the problem specifies.
- Round appropriately – keep extra figures until the final step.
Crossing each item off the list dramatically reduces the chance of a hidden pitfall slipping through It's one of those things that adds up..
Conclusion
Stoichiometric conversions—whether you’re turning 2.43 g of hydrogen peroxide into grams of O₂ or scaling up to industrial batches—rely on a straightforward chain of unit‑consistent steps. The most frequent sources of error are:
- Mismatched molar masses (atom vs. molecule).
- Misreading coefficients in the balanced equation.
- Overlooking a limiting reactant when multiple reactants are present.
- Premature rounding, which erodes accuracy.
By keeping the three‑step workflow (mass → moles → product moles → mass) front‑and‑center, using the quick‑reference tables, and ticking off the checklist, you can manage any oxygen‑mass problem with confidence. Practically speaking, the next time you encounter a mysterious “2. 43” in a chemistry question, you’ll instantly know whether you’re dealing with grams, moles, or something else—and you’ll have the tools to turn that number into a precise, reliable answer. Happy calculating!
Some disagree here. Fair enough.
7. Common “Gotchas” and How to Dodge Them
| Pitfall | Why It Happens | Quick Fix |
|---|---|---|
| Using the atomic mass of O instead of the molecular mass of O₂ | The equation often involves O₂, but the periodic table lists 16.That said, 00 g mol⁻¹ for a single O atom. Worth adding: | Remember: O₂ = 2 × 16. Here's the thing — 00 = 32. That's why 00 g mol⁻¹. Keep a small note on your desk or in the margin of your notebook. Plus, |
| Forgetting to convert °C to K | Gas‑law calculations require absolute temperature. | Add 273.15 to every Celsius temperature before plugging it into the ideal‑gas equation. |
| Mix‑up between volume at STP vs. NTP | Different textbooks define “standard” conditions differently (STP = 0 °C, 1 atm; NTP = 20 °C, 1 atm). | Check the problem statement. If it simply says “standard conditions,” assume 0 °C unless otherwise specified, or explicitly ask the instructor. Also, |
| Assuming 100 % yield | Real reactions rarely go to completion; side reactions, incomplete conversion, and product loss lower the actual yield. Practically speaking, | If the problem gives a percent yield, apply it after you have the theoretical mass. In real terms, |
| Neglecting the stoichiometric coefficient of the product | It’s easy to forget a factor of 2, ½, or 3 when moving from reactant moles to product moles. | Write the mole ratio as a fraction (e.g., ( \frac{2\ \text{mol O₂}}{4\ \text{mol H₂O₂}} )) and keep it visible while you calculate. But |
| Rounding intermediate results | Early rounding can cascade into a large final error. Even so, | Keep at least five significant figures through all intermediate steps; round only in the final answer. |
| Treating gases as liquids | The density of O₂ gas (≈1.43 g L⁻¹ at STP) is far lower than that of liquid O₂ (≈1.14 g mL⁻¹). | Verify the physical state the problem asks for; use the appropriate conversion (ideal‑gas law for gases, density for liquids). |
8. A Mini‑Case Study: From Lab‑Scale to Plant‑Scale
Scenario – A pharmaceutical plant needs to generate 5 tonnes of O₂ per day by decomposing hydrogen peroxide. The reaction is the same as before:
[ 2;\text{H}_2\text{O}_2(l) ;\longrightarrow; 2;\text{H}_2\text{O}(l) + \text{O}_2(g) ]
The plant operates at 25 °C and 1 atm, and the process is 85 % efficient.
Step‑by‑step scaling
| Step | Calculation | Result |
|---|---|---|
| 1️⃣ Convert daily O₂ demand to moles | (5;\text{t} = 5.0\times10^{6};\text{g}) <br> (n_{\text{O₂,req}} = \frac{5.So 0\times10^{6};\text{g}}{32. 00;\text{g mol}^{-1}} = 1.56\times10^{5};\text{mol}) | (1.56\times10^{5}) mol |
| 2️⃣ Adjust for 85 % efficiency | (n_{\text{O₂,theor}} = \frac{n_{\text{O₂,req}}}{0.Here's the thing — 85} = 1. Now, 84\times10^{5};\text{mol}) | (1. 84\times10^{5}) mol |
| 3️⃣ Determine required H₂O₂ moles (1:1 ratio) | (n_{\text{H₂O₂}} = 1.84\times10^{5};\text{mol}) | Same as O₂ |
| 4️⃣ Convert H₂O₂ moles to mass | (m_{\text{H₂O₂}} = n_{\text{H₂O₂}}\times34.02;\text{g mol}^{-1}=6.Which means 26\times10^{6};\text{g}=6. 26;\text{t}) | 6.26 tonnes of H₂O₂ |
| 5️⃣ Check reactor volume (optional) | At 25 °C, 1 atm, ideal‑gas law gives (V_{\text{O₂}} = nRT/P = 1.On top of that, 84\times10^{5}\times0. 08206\times298/1 = 4.5\times10^{6};\text{L}) ≈ 4 500 m³ of O₂ gas per day. |
This is the bit that actually matters in practice Nothing fancy..
Take‑away: By applying the same stoichiometric backbone used for a 2.43‑g sample, we can confidently scale the calculation to multi‑tonne industrial operations, simply by preserving unit consistency and inserting the appropriate efficiency factor.
9. Practice Problems (With Answers)
| # | Problem Statement | Answer (rounded) |
|---|---|---|
| 1 | 12.(C₆H₁₂O₆ + 6 O₂ → 6 CO₂ + 6 H₂O) | 3.Because of that, what volume of O₂ gas is collected at STP? 5 g of Na₂CO₃ is heated to produce CO₂ gas. 74 g |
| 3 | 3.750 g of CaCl₂ reacts with excess Na₂CO₃. Also, how many liters of CO₂ are formed at 25 °C and 1 atm? 6 L | |
| 2 | 0.Worth adding: 00 g | |
| 4 | In a lab, 1. 20 g of H₂O₂ decomposes with a 92 % yield. 68 L | |
| 5 | 25 mL of 0.Still, (Reaction: Na₂CO₃ → Na₂O + CO₂) | 5. In real terms, 200 M H₂SO₄ is neutralized by NaOH. |
Tip: Work through these problems using the three‑step workflow and the checklist above; you’ll see how quickly the process becomes second nature.
10. Final Thoughts
Stoichiometry is the mathematical skeleton that holds chemistry together. Once you internalize the three‑step conversion chain, keep the balancing equation front‑and‑center, and employ the quick‑reference tables, the “2.43‑gram‑of‑something” riddles dissolve into routine arithmetic Easy to understand, harder to ignore..
Remember:
- Units are your compass. Never lose sight of them; they will guide you back when an answer looks odd.
- The limiting reactant is the gatekeeper of product formation—identify it early.
- Yield and equilibrium are reality checks that convert the ideal world of balanced equations into the messy world of real chemistry.
By treating each problem as a short story—introducing the characters (reactants), setting the scene (conditions), following the plot (reaction pathway), and delivering the climax (product amount)—you’ll not only solve the numbers but also develop a deeper intuition for how matter transforms Easy to understand, harder to ignore. Surprisingly effective..
So the next time you see a mass of 2.And 43 g, a pressure of 1. So naturally, 02 atm, or a percent yield of 78 %, you’ll know exactly which page of the stoichiometry playbook to open, and you’ll be ready to write the ending yourself. Happy calculating!
11. Common Pitfalls & How to Dodge Them
| Pitfall | Why It Happens | Quick Fix |
|---|---|---|
| Forgetting to convert temperature to Kelvin | °C is easy to overlook, especially when the problem already supplies a “room‑temperature” hint. That's why | Rule of thumb: *Whenever you see a gas‑law calculation, immediately write “T = ___ °C → ___ K”. Still, * |
| Mixing up molar mass units | Some tables list g mol⁻¹, others kg kmol⁻¹. In real terms, the extra factor of 1 000 can flip your answer by three orders of magnitude. And | Keep a dual‑column cheat sheet: one column for g mol⁻¹ (most organic compounds), another for kg kmol⁻¹ (industrial chemicals). |
| Assuming 100 % yield without checking the problem | Many textbook examples explicitly state “theoretical yield,” but real‑world questions often hide the yield in a footnote. In real terms, | Scan the whole statement first; underline any percentage, “actual”, “recovered”, or “efficiency”. |
| Using the wrong gas constant | R = 0.08206 L·atm·mol⁻¹·K⁻¹ works for L‑atm, while R = 8.314 J·mol⁻¹·K⁻¹ is needed for Pa·m³. | Write the units of R on your worksheet before plugging numbers. If the problem mixes L and Pa, convert everything to a single system first. Practically speaking, |
| Neglecting the limiting reactant when excess is present | It’s easy to calculate product from the first reactant you see and forget the second one. That's why | After you balance the equation, list moles of every reactant side‑by‑side and compare the stoichiometric ratios. The smallest ratio = limiting. |
12. A Mini‑Calculator in Your Head
If you’re short on paper or a calculator, these mental‑math shortcuts can shave seconds off each step:
| Operation | Approximation | When to Use |
|---|---|---|
| Moles ≈ mass / 50 | Works for many common organic compounds (C ≈ 12, H ≈ 1, O ≈ 16 → average ≈ 30–40; 50 is a safe over‑estimate). | Quick sanity check: “Is my answer within a factor of 2?” |
| Gas volume at STP ≈ 22 L · moles | Exact value is 22.But 414 L, but 22 L is easier to remember. | Estimating the order of magnitude of a gas‑law answer. |
| Percent yield ≈ (actual / theoretical) × 100 | If you know the theoretical mass is 5.0 g and you measured 4.Also, 2 g, mental division 4. 2/5 ≈ 0.84 → 84 %. | When you need a rough answer for a lab report draft. Day to day, |
| Temperature correction factor | ΔT (°C) → ΔT (K) is identical, so 25 °C → 298 K ≈ 300 K for back‑of‑the‑envelope calculations. | When you only need a ball‑park volume of a gas. |
13. Beyond the Basics: When Stoichiometry Meets Real Chemistry
| Scenario | Extra Consideration | How to Incorporate It |
|---|---|---|
| Reversible reactions (e.Practically speaking, g. Because of that, , Haber process) | Equilibrium constants (Kₚ, K_c) dictate how much product actually forms. That said, | Perform an ICE table (Initial‑Change‑Equilibrium) after the stoichiometric step, then solve for equilibrium concentrations. |
| Multiphase systems (solid + gas) | Solids and pure liquids have activity ≈ 1, so they drop out of the equilibrium expression. | Focus the equilibrium calculation on the gaseous or aqueous species; use stoichiometry to relate them to the solid. |
| Catalysis | Catalysts change the rate, not the stoichiometry. But | No extra calculation needed; just note the catalyst in the reaction description. Plus, |
| Polymerization | Degree of polymerization (DP) determines how many monomer units join. | Convert monomer moles → polymer mass using DP × molar mass of repeat unit. Think about it: |
| Redox balancing in acidic/basic media | Electrons must be balanced in half‑reactions before combining. | Use the half‑reaction method, then proceed with the usual mole‑to‑mass conversions. |
14. A Quick‑Reference “One‑Page” Summary
Below is a printable cheat sheet you can tape to the inside of your lab notebook.
| Step | Action | Key Equation / Note |
|---|---|---|
| 1️⃣ | Write & balance the chemical equation. | Verify atoms & charge. That's why <br>Gas volume: (V = n·\frac{RT}{P}). Now, |
| 2️⃣ | Convert given quantities → moles. | |
| 4️⃣ | Use stoichiometric ratios to find theoretical moles of each product. Now, | (n_{\text{product}} = n_{\text{lim}}·\frac{\nu_{\text{product}}}{\nu_{\text{lim}}}). |
| 3️⃣ | Identify the limiting reactant. | |
| 5️⃣ | Convert theoretical moles → desired units (mass, volume, pressure). | |
| 7️⃣ | Check units and significant figures. That said, | |
| 6️⃣ | Apply percent yield (if given). Which means | “Mass of CuSO₄·5H₂O formed = 3. |
| 8️⃣ | Write the final answer with a clear label. | Compare (n_{\text{actual}}/ \nu_{\text{stoich}}). |
This is where a lot of people lose the thread.
Conclusion
Stoichiometry may initially feel like a maze of symbols, but once the three‑step workflow—convert, compare, convert back—is ingrained, each problem resolves itself in a matter of minutes. By anchoring every calculation to a balanced equation, vigilantly tracking units, and remembering the limiting‑reactant rule, you transform a seemingly abstract algebraic exercise into a reliable, repeatable laboratory tool.
The examples above, from a modest 2.In real terms, 43 g sample to a multi‑tonne industrial process, demonstrate that the same fundamental principles scale without loss of accuracy—provided you stay disciplined about unit consistency and efficiency factors. The practice problems reinforce the workflow, while the pitfalls, mental shortcuts, and extensions into equilibrium and redox chemistry arm you for the diverse scenarios you’ll encounter in coursework, research, or industry But it adds up..
In short, stoichiometry is the language of chemical change; mastering its grammar lets you read, write, and translate any reaction story with confidence. On top of that, keep the cheat sheet handy, work through a few problems each week, and soon the calculations that once seemed daunting will become second nature. Happy reacting!
Real talk — this step gets skipped all the time That alone is useful..
15. Beyond the Basic Reaction: Stoichiometry in Multi‑Step Pathways
In many real‑world scenarios the reaction you’re asked to analyze isn’t a single step but a cascade of inter‑linked transformations. Whether you’re working with a catalytic cycle, a biotransformation, or a multi‑product synthesis, the same stoichiometric logic applies—just with a few extra layers of bookkeeping And it works..
15.1 Cascades and “Net” Equations
When a reagent is consumed in one step and regenerated in another, the intermediate’s stoichiometry can be eliminated by adding the equations together. This is often called a net reaction.
Example – Catalytic oxidation of ethanol to acetaldehyde (C₂H₅OH → CH₃CHO)
| Step | Reaction | Coefficient | Notes |
|---|---|---|---|
| 1 | ( \text{C}_2\text{H}_5\text{OH} + \tfrac12 \text{O}_2 \rightarrow \text{CH}_3\text{CHO} + \tfrac12 \text{H}_2\text{O} ) | 1 | Primary oxidation |
| 2 | ( \text{CH}_3\text{CHO} + \tfrac12 \text{O}_2 \rightarrow \text{CH}_3\text{COOH} ) | 1 | Secondary oxidation |
Add the two steps:
[ \text{C}_2\text{H}_5\text{OH} + \text{O}_2 \rightarrow \text{CH}_3\text{COOH} + \tfrac12 \text{H}_2\text{O} ]
Now you can treat the whole process as a single reaction for yield calculations, provided the intermediate doesn’t accumulate Still holds up..
15.2 Redox Balancing with Electron Transfer
In redox reactions the stoichiometric coefficients are often dictated by electron balance rather than atom balance alone. The half‑reaction method (already mentioned in the cheat sheet) is indispensable And it works..
Quick reminder – Oxidation half‑reaction gives the number of electrons lost; reduction half‑reaction gives electrons gained. Multiply each by the appropriate factor so the electrons cancel, then add the two halves.
15.3 Stoichiometry in Equilibrium Systems
When a reaction is reversible, the extent of reaction (ξ) becomes the variable of interest. The stoichiometric coefficients still dictate the relationship between concentrations at any point:
[ [\text{A}] = [\text{A}]0 - \nu{\text{A}}\xi,\quad [\text{B}] = [\text{B}]0 - \nu{\text{B}}\xi,\quad [\text{C}] = [\text{C}]0 + \nu{\text{C}}\xi ]
Plugging these into the equilibrium constant expression (K = \frac{[\text{C}]^{\nu_{\text{C}}}}{[\text{A}]^{\nu_{\text{A}}}[\text{B}]^{\nu_{\text{B}}}}) allows you to solve for ξ, and from there the actual amounts of each species at equilibrium.
16. Common Mistakes and How to Spot Them
| Mistake | Why it Happens | Quick Fix |
|---|---|---|
| Using grams instead of moles | Confusing mass with quantity | Always convert to moles before applying stoichiometric ratios |
| Ignoring the limiting reactant | Overlooking that excess reagent doesn’t affect product | Check (n_{\text{actual}}/\nu_{\text{stoich}}) for each reactant |
| Mixing unit systems | Switching between SI and CGS mid‑calculation | Stick to one system; convert at the very beginning |
| Rounding too early | Losing precision | Keep full‑precision intermediate values; round only final answer |
| Assuming 1 L of any gas = 1 mol | Temperature/pressure not standard | Use (n = PV/RT) for real conditions |
| Forgetting to include water of crystallization | Ignoring hydrates in solubility calculations | Include the hydrate’s formula weight in the molar mass |
| Treating percent yield as 100 % | Overlooking side reactions | Always apply reported yield unless stated otherwise |
17. Practice Problems – Test Your Skills
-
Industrial Scale – A plant converts 5 Mt of methane to methanol using a catalytic partial oxidation process:
[ \text{CH}_4 + \tfrac12 \text{O}_2 \rightarrow \text{CH}_3\text{OH} ]
If the overall efficiency is 80 %, how many kilograms of methanol are produced? -
Redox Balancing – Balance the reaction in acidic solution:
[ \text{Cr}_2\text{O}_7^{2-} + \text{C}_2\text{O}_4^{2-} \rightarrow \text{Cr}^{3+} + \text{CO}_2 ] -
Equilibrium – For the reaction ( \text{N}_2 + 3\text{H}_2 \rightleftharpoons 2\text{NH}_3 ) at 500 K, the equilibrium constant (K_p = 0.1). If 1.0 mol of ( \text{N}_2 ) and 3.0 mol of ( \text{H}_2 ) are mixed in a 10 L vessel at 1 atm, calculate the equilibrium partial pressure of ammonia.
-
Limiting Reactant with a Catalyst – 2.5 g of ( \text{NaNO}_3 ) reacts with excess ( \text{KOH} ) in a catalytic decomposition:
[ 2\text{NaNO}_3 \rightarrow 2\text{NaNO}_2 + \text{O}_2 ]
What is the theoretical yield of ( \text{O}_2 ) in liters at 25 °C and 1 atm? -
Real‑World Application – In a wastewater treatment plant, 150 L of a solution containing 1.2 g L(^{-1}) of ( \text{Na}_2\text{S}_2\text{O}_3 ) is treated with excess ( \text{H}_2\text{O}_2 ) to convert thiosulfate to sulfate. The reaction is:
[ \text{Na}_2\text{S}_2\text{O}_3 + \text{H}_2\text{O}_2 \rightarrow \text{Na}_2\text{SO}_4 + \text{S} + \text{O}_2 ]
Assuming 100 % conversion, how many moles of elemental sulfur are produced?
(Answers are provided in the supplementary PDF.)
18. Resources for Further Exploration
-
Textbooks
- Physical Chemistry by Peter Atkins & Julio De Paula – Chapter 3, “Stoichiometry and the Mole Concept”
- Quantitative Chemical Analysis by Daniel C. Harris – Section on “Stoichiometric Calculations”
-
Online Calculators
- Stoichiometry Calculator (ChemCalc.org) – Input reactants and desired product to get all intermediate values.
- Reaction Balancer (WebMO, PSE) – Offers half‑reaction balancing and redox checks.
-
Software
- ChemDraw – Visualize balanced equations and automatically generate stoichiometric tables.
- MATLAB / Python (SciPy) – For large‑scale stoichiometric matrices and sensitivity analyses.
-
Tutorial Videos
- Khan Academy – “Stoichiometry” playlist (15‑minute segments).
- MIT OpenCourseWare – “Chemical Thermodynamics” (focus on equilibrium and reaction quotients).
19. Final Thoughts
Stoichiometry is the backbone of quantitative chemistry. Whether you’re a student tackling textbook exercises, a researcher optimizing a synthetic route, or an engineer scaling a production line, the ability to translate a balanced equation into real‑world quantities is indispensable. By mastering the core workflow—converting to moles, identifying the limiting reactant, applying stoichiometric ratios, and converting back to the desired units—you reach a powerful toolkit that transcends every chemical discipline.
Remember: the key to fluency is practice. Keep a running log of solved problems, revisit the cheat sheet when you’re in a hurry, and never skip the unit check. Over time, the calculations will feel less like algebraic gymnastics and more like a natural extension of your chemical intuition That's the part that actually makes a difference..
Happy calculating, and may your reactions always go to completion!