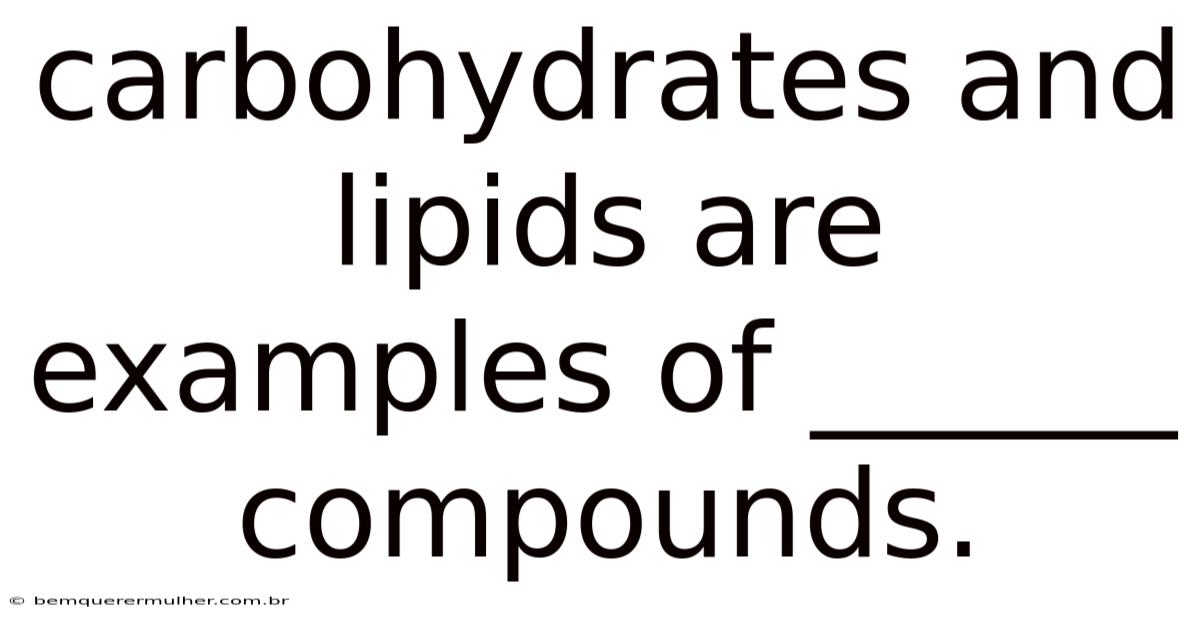Carbohydrates and lipids are examples of organic compounds. Day to day, this fundamental classification underpins much of biochemistry and dictates the roles these molecules play in living organisms. So while the statement seems simple, it opens a door to a complex and fascinating world of carbon-based chemistry that is essential for life. Understanding what makes a compound organic, and how carbohydrates and lipids fit into this category, provides a crucial foundation for grasping biological processes, energy metabolism, and the structural integrity of cells It's one of those things that adds up. Less friction, more output..
No fluff here — just what actually works.
Introduction to Organic Chemistry in Biology
The term organic in a chemical context has a specific meaning that differs from its everyday usage. In real terms, today, organic chemistry is defined as the study of carbon-containing compounds. Also, this theory was debunked in the 19th century when chemists successfully synthesized organic compounds like urea from inorganic starting materials. Historically, organic chemistry was the study of compounds derived from living organisms, leading to the belief that a "vital force" was necessary to create them. Think about it: the key element is carbon, due to its unique ability to form four stable covalent bonds with other atoms, including other carbon atoms. This property allows for the creation of long chains, rings, and incredibly diverse molecular structures.
Carbohydrates and lipids are examples of organic compounds because they both contain carbon, hydrogen, and oxygen atoms bonded together in specific configurations. While they share this basic classification, their distinct chemical structures lead to vastly different physical properties and biological functions. Carbohydrates are generally more polar and soluble in water, making them ideal for quick energy transport and structural support. Lipids, on the other hand, are largely nonpolar and hydrophobic, which makes them perfect for long-term energy storage and as the primary building blocks of cellular membranes. Exploring these differences reveals the elegant diversity within the realm of organic molecules It's one of those things that adds up..
The Structural and Functional Diversity of Carbohydrates
Carbohydrates, often referred to as saccharides, are perhaps the most abundant organic compounds on Earth. These molecules are highly soluble in water due to the presence of numerous hydroxyl (-OH) groups, which can form hydrogen bonds with water molecules. They serve as a primary energy source for most life forms and play critical roles in cell recognition and signaling. The simplest carbohydrates are monosaccharides, such as glucose and fructose, which are single sugar units with the general formula (CH₂O)ₙ. This solubility is why glucose is the preferred form of sugar transported in the blood of animals and used directly by cells for energy production through cellular respiration.
The importance of carbohydrates and lipids are examples of organic compounds is highlighted when we compare their structures. While lipids are characterized by long hydrocarbon chains that repel water, carbohydrates are hydrophilic. Which means this structural difference dictates their function. Complex carbohydrates, or polysaccharides, are formed when monosaccharides link together through dehydration synthesis reactions. Examples include:
- Starch: A storage polysaccharide in plants, composed of amylose and amylopectin.
- Glycogen: The storage polysaccharide in animals, primarily found in the liver and muscles.
- Cellulose: A structural polysaccharide that forms the cell walls of plants, providing rigidity and strength.
Not obvious, but once you see it — you'll see it everywhere Nothing fancy..
These diverse structures arise from the same basic building blocks but are arranged differently, showcasing the versatility of organic chemistry. The hydroxyl groups on carbohydrates also make them reactive participants in many biochemical pathways, acting as intermediates in metabolism and forming glycosidic bonds with other molecules like lipids and proteins to create glycolipids and glycoproteins, which are crucial for cell membrane function.
The Hydrophobic World of Lipids
In stark contrast to carbohydrates, lipids are a diverse group of organic compounds that are largely hydrophobic or "water-fearing.Because of that, they include fats, oils, waxes, phospholipids, and steroids. Practically speaking, " This property stems from their nonpolar carbon-hydrogen bonds, which do not interact favorably with polar water molecules. Lipids are not a single type of molecule but rather a category defined by their solubility behavior. Despite this diversity, their primary roles revolve around energy storage, insulation, and membrane structure.
The most well-known lipids are triglycerides, which consist of a glycerol backbone attached to three fatty acid chains. These molecules are an incredibly efficient form of energy storage. Because they are hydrophobic, they can be packed tightly together in anhydrous environments (without water), storing a large amount of energy in a compact form. When the body requires energy, these triglycerides are broken down through a process called beta-oxidation, releasing fatty acids that can be used to generate ATP.
Another critical class of lipids is phospholipids. Consider this: these molecules are amphipathic, meaning they have both a hydrophobic tail and a hydrophilic head. In an aqueous environment, phospholipids spontaneously arrange themselves into a bilayer, with the hydrophobic tails facing inward and the hydrophilic heads facing outward toward the water. This structure forms the foundation of all cellular membranes, creating a selective barrier that controls the movement of substances in and out of the cell. This dual nature is the fundamental reason why lipids are examples of organic compounds that build cellular membranes. Cholesterol, another lipid, is also embedded within this bilayer, modulating membrane fluidity and stability Simple, but easy to overlook..
The Metabolic Interplay Between Carbohydrates and Lipids
While carbohydrates and lipids are examples of organic compounds with distinct structures, they are not isolated in the body's metabolism. They exist in a dynamic interplay, particularly concerning energy balance. Day to day, when carbohydrate intake exceeds immediate energy needs, the body has the capacity to convert these excess carbohydrates into fatty acids and subsequently into triglycerides for storage in adipose tissue. This process is a key mechanism for long-term energy storage.
No fluff here — just what actually works Worth keeping that in mind..
Conversely, during periods of fasting or intense exercise when carbohydrate reserves are depleted, the body can mobilize its fat stores. That's why through lipolysis, triglycerides are broken down into glycerol and fatty acids. That said, the fatty acids undergo beta-oxidation to produce acetyl-CoA, which enters the Krebs cycle to generate ATP. Plus, this metabolic flexibility underscores the importance of both types of organic compounds in maintaining homeostasis. The glycerol can be converted into glucose via gluconeogenesis, providing a vital energy source for the brain and red blood cells. The body preferentially uses carbohydrates for immediate energy but relies on lipids for sustained energy needs, demonstrating a sophisticated biological economy.
Debunking Myths and Understanding Classification
A common point of confusion arises when discussing the classification of carbohydrates and lipids are examples of organic compounds. Plus, they are critical for hormone production (e. g.While excessive intake of certain types of lipids, particularly saturated and trans fats, can contribute to health issues, lipids are essential for life. Some people mistakenly believe that because lipids are fats, they are inherently unhealthy. , steroid hormones like testosterone and estrogen), vitamin absorption (vitamins A, D, E, and K are fat-soluble), and brain function And it works..
Similarly, not all carbohydrates are "bad." Complex carbohydrates found in whole grains, vegetables, and legumes provide sustained energy, dietary fiber, and essential nutrients. Here's the thing — the key is understanding the difference between simple sugars and complex polysaccharides, and between healthy and unhealthy fats. The organic nature of these compounds means they are subject to the same chemical principles, but their biological outcomes are determined by their specific structures and how the body processes them Not complicated — just consistent..
Frequently Asked Questions
Q1: Why is carbon so important in defining organic compounds? Carbon's tetravalent nature allows it to form stable covalent bonds with up to four other atoms. This enables the formation of long chains, branched structures, and rings, creating the molecular complexity necessary for life. No other element can build such a diverse array of stable molecules, which is why carbon is the backbone of all organic compounds, including carbohydrates and lipids.
Q2: Are all organic compounds energy-rich? No, not all organic compounds are high in energy. While lipids are highly energy-dense, carbohydrates provide a quicker, more readily accessible form of energy. Structural organic compounds like cellulose, while organic, are not used for energy production in humans because we lack the enzymes to break them down. The energy content is a property specific to the molecule's structure and its bonds Practical, not theoretical..
Q3: Can the body make all the lipids it needs? The body can synthesize most lipids from carbohydrates and proteins through metabolic pathways. Still, there are certain essential fatty acids, like linoleic acid and alpha-linolenic acid, that the body cannot produce and must be obtained from the diet. These are the "essential fats" crucial for maintaining health.
**Q4: How do carbohydrates and lipids differ in their solubility
Understanding the distinctions between carbohydrates and lipids further clarifies why misinformation about their health impacts persists. And carbohydrates are generally water-soluble, making them more easily absorbed and utilized for immediate energy needs. Because of that, in contrast, lipids are typically fat-soluble, allowing them to be stored in adipose tissue and released slowly over time. This solubility difference explains their varied roles in the body: carbohydrates fuel cells and support daily functions, while lipids are vital for insulation, hormone synthesis, and long-term energy storage.
No fluff here — just what actually works.
This contrast underscores the importance of balance in nutrition. While many associate high-fat foods with poor health, it's essential to recognize that the type of fat matters more than quantity alone. Unsaturated fats, found in nuts, seeds, and fish, are beneficial, whereas saturated and trans fats, often from processed foods, can negatively affect cardiovascular health. Similarly, complex carbohydrates from fruits and vegetables provide sustained energy and essential nutrients, contrasting with simple sugars that cause rapid spikes in blood glucose.
The body's ability to adapt to these differences highlights the significance of informed dietary choices. That said, by appreciating the unique functions of both carbohydrates and lipids, individuals can make better decisions that align with their health goals. It also emphasizes how scientific understanding helps dispel myths and promote a more nuanced view of nutrition Small thing, real impact..
At the end of the day, the classification of organic compounds like carbohydrates and lipids is fundamental to grasping their roles in human biology. Which means recognizing their structural and functional differences empowers individuals to make choices that support overall well-being, reinforcing the value of continuous learning in scientific literacy. Embracing this knowledge allows us to move beyond outdated beliefs and support healthier lifestyles And it works..