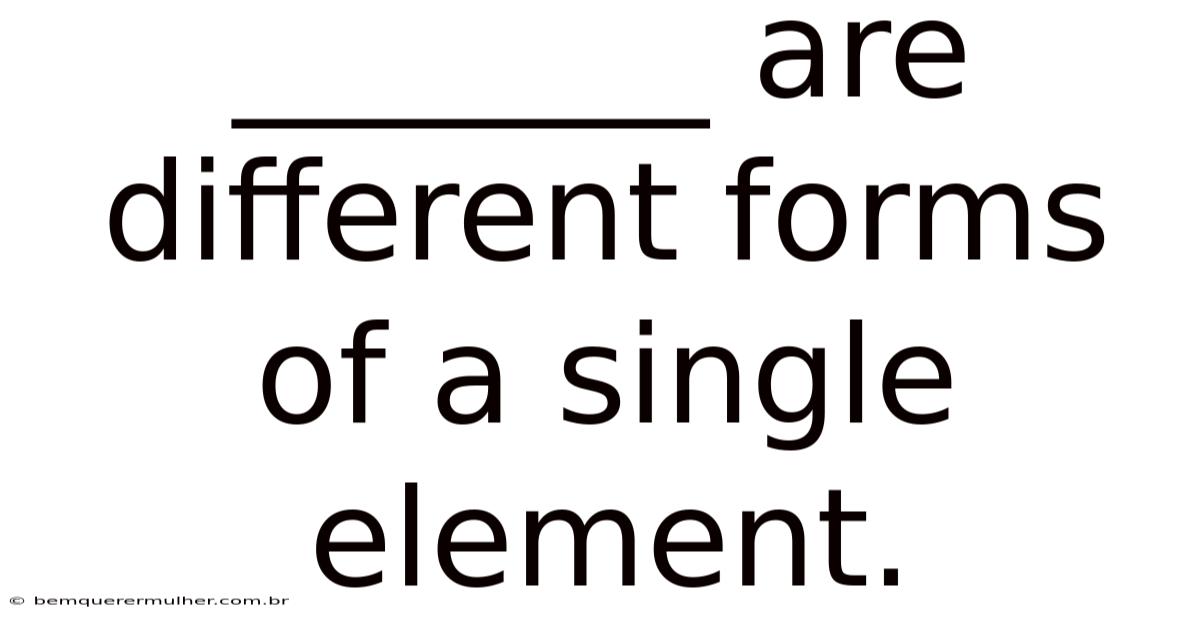Allotropes are different formsof a single element. Day to day, this fascinating phenomenon highlights the incredible versatility of atoms and the profound impact their arrangement can have on the properties of a substance. Understanding allotropes is crucial not only for grasping fundamental chemistry but also for appreciating the diverse materials that shape our world, from the graphite in your pencil to the diamond on a ring. Let's dig into the world of allotropes, exploring their definition, how they differ, and why they matter The details matter here..
The official docs gloss over this. That's a mistake.
What Exactly Are Allotropes?
At its core, an allotrope represents a distinct structural modification of an element. It occurs when atoms of that single element bond together in different ways, leading to variations in physical and chemical properties. Still, think of it as the element wearing different outfits; the underlying material is the same, but how it's put together results in vastly different appearances and behaviors. This ability to exist in multiple structural forms is known as allotropy Most people skip this — try not to..
How Do Allotropes Differ?
The key difference between allotropes lies in their atomic bonding and crystal structure. While the chemical composition (the element itself) remains identical, the arrangement of atoms dictates how they interact. This arrangement can be influenced by factors like temperature, pressure, and the presence of catalysts.
- Diamond vs. Graphite (Carbon): Carbon is a classic example. Diamond forms under immense pressure deep within the Earth, where each carbon atom is bonded covalently to four others in a rigid, three-dimensional tetrahedral lattice. This structure makes diamond incredibly hard and transparent. In stark contrast, graphite forms under lower pressure conditions, where carbon atoms bond covalently in flat, hexagonal sheets stacked loosely on top of each other. These weak interlayer forces allow the sheets to slide easily, making graphite soft and slippery, perfect for pencils. Graphene, a single layer of graphite, is an even more extreme allotrope, incredibly strong and conductive.
- O₂ vs. O₃ (Oxygen): Oxygen exists as two common allotropes: dioxygen (O₂), the vital gas we breathe, and ozone (O₃), a powerful oxidizing agent found in the upper atmosphere. In O₂, two oxygen atoms share two pairs of electrons. In O₃, three oxygen atoms share electrons in a resonance structure, resulting in a bent molecule with a distinct odor and different reactivity.
- White Phosphorus vs. Red Phosphorus (Phosphorus): Phosphorus has several allotropes. White phosphorus is highly reactive, waxy, and ignites spontaneously in air. Red phosphorus is much more stable, less toxic, and used in safety matches. The difference stems from the molecular structure: white phosphorus consists of P₄ molecules, while red phosphorus has a more complex, polymeric structure.
- Buckminsterfullerene (C₆₀) vs. Diamond (Carbon): This is another carbon example. Buckminsterfullerene, or "buckyball," consists of 60 carbon atoms arranged in a hollow, soccer-ball-like cage. This structure gives it unique electronic and chemical properties, distinct from both diamond and graphite.
Why Do Allotropes Exist?
Allotropes arise due to the different ways atoms can achieve the most stable electron configuration (lowest energy state) under varying environmental conditions. The specific bonding arrangement that minimizes energy can change with temperature, pressure, or the presence of impurities. Worth adding: for carbon, the high pressure and temperature deep underground favor the formation of the dense, tetrahedral lattice of diamond. At the Earth's surface, lower pressure allows carbon to form the layered structure of graphite. Similarly, ozone forms in the upper atmosphere when solar radiation breaks apart O₂ molecules, allowing the freed atoms to recombine into O₃, which is less stable at lower altitudes And that's really what it comes down to..
This changes depending on context. Keep that in mind That's the part that actually makes a difference..
Scientific Explanation: The Role of Bonding and Structure
The properties of an allotrope are fundamentally dictated by its crystal structure and bonding type:
- Bonding: Allotropes often involve different types of bonding. Diamond and graphite are both covalent networks. Oxygen allotropes involve covalent bonding. Phosphorus allotropes involve covalent bonding in the molecular form (white P) and a mix of covalent and ionic bonding in the polymeric form (red P).
- Crystal Structure: This is critical. The specific geometric arrangement of atoms within the crystal lattice determines density, hardness, melting point, electrical conductivity, and optical properties. Diamond's tetrahedral lattice is extremely rigid, while graphite's layered structure allows slippage. The cage structure of C₆₀ allows for unique electronic properties.
- Phase Transitions: Allotropes can interconvert under specific conditions. As an example, graphite can slowly convert to diamond under extremely high pressure and temperature over geological timescales. Similarly, red phosphorus can convert to white phosphorus if heated rapidly.
FAQ: Common Questions About Allotropes
- Q: Are allotropes different compounds? No. Allotropes are different forms of the same element. Compounds involve different elements chemically bonded together.
- Q: Can any element have allotropes? Most elements with multiple stable bonding configurations can exhibit allotropy, especially those in the p-block (like carbon, phosphorus, sulfur) and some transition metals (like iron). Elements with simple atomic structures like helium or neon typically do not form allotropes.
- Q: Why are allotropes important? Allotropes demonstrate the profound impact of atomic arrangement on material properties. This knowledge is crucial for developing new materials: diamond for cutting tools and electronics, graphene for ultra-strong composites and sensors, carbon nanotubes for nanotechnology, and various phosphorus allotropes for fertilizers and matches. Understanding allotropy helps scientists tailor materials for specific applications.
- Q: How do we identify allotropes? Allotropes are identified through techniques like X-ray crystallography (to determine crystal structure), spectroscopy (to analyze bonding and electronic structure), and physical property measurements (density, hardness, melting point, conductivity).
- Q: Can allotropes have different chemical reactivity? Yes! While the element's fundamental chemistry remains the same, the different atomic arrangements can expose atoms to different environments within the structure. Take this: the reactive white phosphorus is far more hazardous than the stable red phosphorus due to its molecular structure.
Conclusion: The Diverse Faces of Matter
Allotropes are a testament to the dynamic nature of matter at the atomic level. They reveal how the same atoms, when arranged differently, can manifest as substances with seemingly contradictory properties – from the hardest known material (diamond) to one of the softest (graphite). Which means this phenomenon isn't just a curious scientific footnote; it's a cornerstone of materials science and chemistry. By understanding and harnessing the different allotropes of elements like carbon, phosphorus, and oxygen, we tap into a vast array of materials that power our technology, shape our tools, and deepen our comprehension of the physical world.
This is the bit that actually matters in practice.
Conclusion: The Diverse Faces of Matter
Allotropes are a testament to the dynamic nature of matter at the atomic level. They reveal how the same atoms, when arranged differently, can manifest as substances with seemingly contradictory properties – from the hardest known material (diamond) to one of the softest (graphite). This phenomenon isn't just a curious scientific footnote; it's a cornerstone of materials science and chemistry. Which means by understanding and harnessing the different allotropes of elements like carbon, phosphorus, and oxygen, we tap into a vast array of materials that power our technology, shape our tools, and deepen our comprehension of the physical world. The next time you see a diamond sparkle or write with a pencil, remember you're witnessing the remarkable diversity hidden within a single element. The ongoing exploration of allotropes promises even more exciting discoveries in the future, continually pushing the boundaries of what's possible and reminding us of the incredible complexity and beauty of the universe we inhabit.