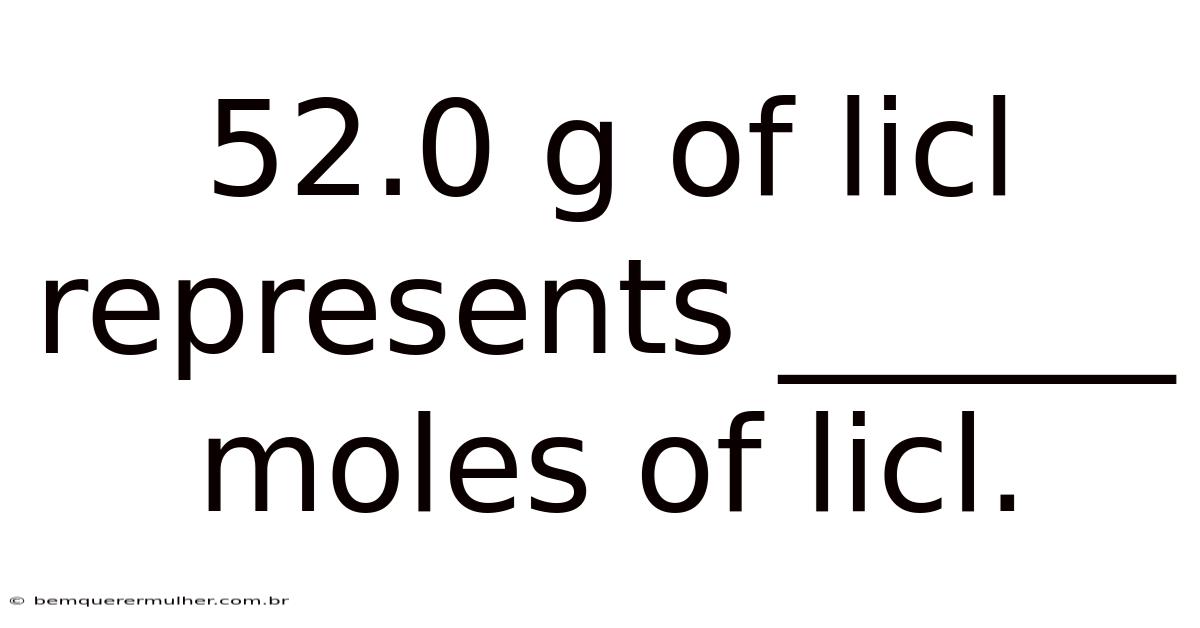How to Calculate the Number of Moles in 52.0 g of LiCl: A Step-by-Step Guide
Chemistry often requires converting between mass and moles to understand the quantity of substances involved in reactions. 0 g of LiCl** (lithium chloride), a common ionic compound used in industrial and laboratory settings. That said, one of the most fundamental skills in stoichiometry is determining how many moles of a compound correspond to a given mass. In this article, we’ll explore how to calculate the number of moles in **52.By breaking down the process into clear steps, we’ll demystify the relationship between mass, molar mass, and moles The details matter here..
Understanding the Basics: Moles and Molar Mass
Before diving into the calculation, let’s clarify the key terms:
- Mole (mol): A unit representing 6.Which means it’s used to count atoms, molecules, or ions in chemistry. 022 × 10²³ particles (Avogadro’s number). - Molar Mass (g/mol): The mass of one mole of a substance, calculated by summing the atomic masses of its constituent elements.
Honestly, this part trips people up more than it should.
For ionic compounds like LiCl, the molar mass is the sum of the atomic masses of lithium (Li) and chlorine (Cl). These values are found on the periodic table:
- Lithium (Li): 6.94 g/mol
- Chlorine (Cl): 35.
Adding these gives the molar mass of LiCl:
**6.94 g/mol + 35.45 g/mol = 42.
This means 1 mole of LiCl weighs 42.39 grams Simple, but easy to overlook..
Step-by-Step Calculation: From Grams to Moles
To find how many moles are in 52.0 g of LiCl, we use the formula:
Moles = Mass (g) / Molar Mass (g/mol)
Step 1: Identify the given values
- Mass of LiCl = 52.0 g
- Molar mass of LiCl = 42.39 g/mol
Step 2: Plug values into the formula
Moles = 52.0 g / 42.39 g/mol
Step 3: Perform the division
Using a calculator:
52.0 ÷ 42.39 ≈ 1.226 moles
Final Answer:
52.0 g of LiCl represents 1.226 moles of LiCl.
Scientific Explanation: Why This Matters
The conversion between mass and moles is critical in chemistry because reactions occur at the molecular level. This leads to for example, if a reaction requires 2 moles of LiCl, knowing the molar mass allows chemists to measure the exact mass (84. 78 g) needed. This precision ensures reactions proceed efficiently and safely No workaround needed..
LiCl itself is a versatile compound. Because of that, - Pharmaceuticals (as a precursor in drug synthesis). It’s used in:
- Battery electrolytes (due to its high ionic conductivity).
- Industrial processes (like oil drilling fluids).
Understanding its molar mass helps professionals optimize these applications It's one of those things that adds up..
Common Questions About Molar Mass and Conversions
Q1: Why do we use molar mass instead of just grams?
A: Molar mass links the macroscopic world (grams) to the microscopic world (atoms/molecules). Reactions depend on the number of particles, not their mass.
Q2: What if the mass has more decimal places?
A: Always use the molar mass with the same number of decimal places as the given mass. To give you an idea, if the mass is 52.00 g (four sig figs), use a molar mass like 42.39 g/mol (four sig figs) Easy to understand, harder to ignore..
Q3: Can molar mass be negative?
A: No. Molar mass is a positive value derived from atomic masses, which are always positive.
Q4: How does temperature affect molar mass?
A: Molar mass is a fixed property of a substance and does not change with temperature or pressure And that's really what it comes down to..
Real-World Applications of LiCl Molar Mass
- Battery Technology:
LiCl is a key component