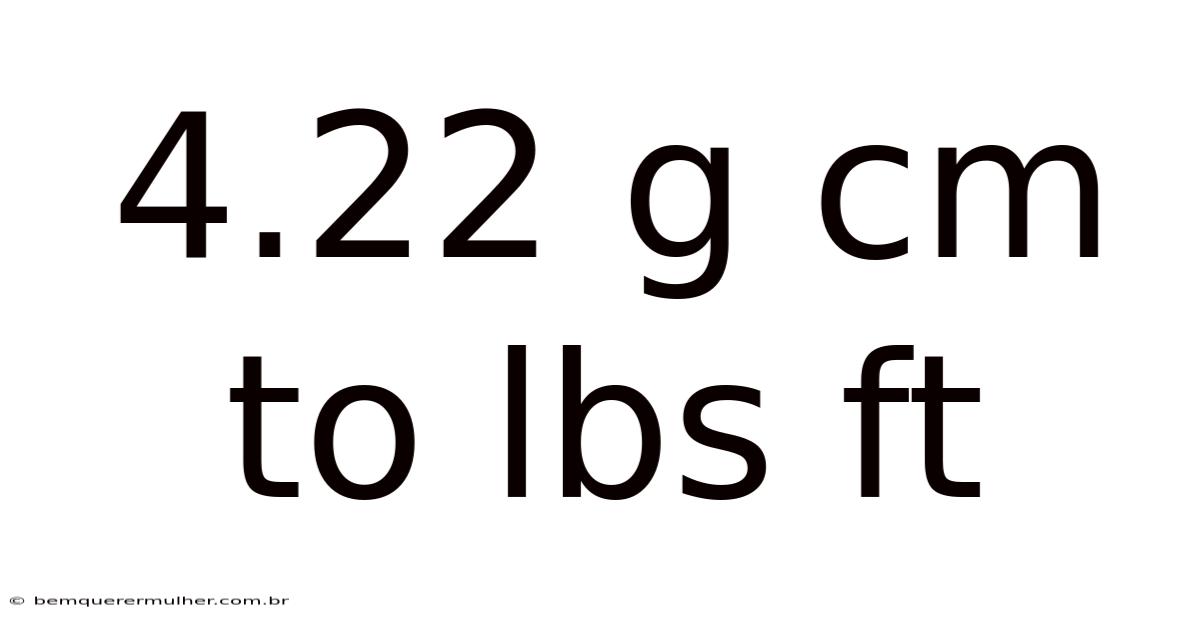Converting 4.22 g cm⁻³ to lbs ft⁻³: A Step‑by‑Step Guide
When working with materials science, engineering, or even everyday calculations, you’ll often encounter density values expressed in grams per cubic centimeter (g cm⁻³). On the flip side, in many engineering contexts—especially in the United States—densities are given in pounds per cubic foot (lbs ft⁻³). Knowing how to convert between these two systems is essential for accurate design, cost estimation, and material selection Turns out it matters..
Below is a comprehensive, easy‑to‑follow guide that explains the theory, the math, and practical tips for converting a density of 4.22 g cm⁻³ into its equivalent in lbs ft⁻³ And it works..
Why the Conversion Matters
- Design and Simulation: Finite element models often require densities in lbs ft⁻³.
- Material Procurement: Suppliers in the U.S. quote prices per pound, so you need the weight per volume in lbs ft⁻³.
- Regulatory Compliance: Some safety standards specify limits in imperial units.
- Cross‑Disciplinary Communication: Engineers, physicists, and chemists may use different unit systems; conversion ensures everyone speaks the same language.
1. Understanding the Units
| Unit System | Symbol | Meaning |
|---|---|---|
| SI (Metric) | g cm⁻³ | Grams per cubic centimeter |
| Imperial | lbs ft⁻³ | Pounds per cubic foot |
The conversion requires two main relationships:
- Mass: 1 gram = 0.00220462 pounds (lb).
- Volume: 1 cubic centimeter = 0.0000353147 cubic feet (ft³).
Combining these gives the conversion factor from g cm⁻³ to lbs ft⁻³.
2. Deriving the Conversion Factor
-
Start with the definition
[ 1\ \text{g cm}^{-3} = \frac{1\ \text{g}}{1\ \text{cm}^3} ] -
Convert the numerator (mass)
[ 1\ \text{g} \times 0.00220462\ \frac{\text{lb}}{\text{g}} = 0.00220462\ \text{lb} ] -
Convert the denominator (volume)
[ 1\ \text{cm}^3 \times 0.0000353147\ \frac{\text{ft}^3}{\text{cm}^3} = 0.0000353147\ \text{ft}^3 ] -
Divide the converted mass by the converted volume
[ \frac{0.00220462\ \text{lb}}{0.0000353147\ \text{ft}^3} \approx 62.4279\ \frac{\text{lb}}{\text{ft}^3} ]
Thus, 1 g cm⁻³ ≈ 62.4279 lbs ft⁻³.
3. Applying the Factor to 4.22 g cm⁻³
Now that we know the factor, the conversion is straightforward:
[ 4.22\ \text{g cm}^{-3} \times 62.4279\ \frac{\text{lb}}{\text{ft}^3\ \text{per g cm}^{-3}} = 263 Turns out it matters..
Rounded to one decimal place: 263.9 lbs ft⁻³.
4. Practical Example: Metal Density
Suppose you’re evaluating a new alloy with a density of 4.22 g cm⁻³ for a structural component:
-
Compute lbs ft⁻³
[ 4.22\ \text{g cm}^{-3} \rightarrow 263.9\ \text{lb ft}^{-3} ] -
Determine Weight of a Cubic Foot
A 1 ft³ block of this alloy weighs 263.9 lb. -
Scale to Design Geometry
If the component is 2 ft × 0.5 ft × 0.25 ft (volume = 0.25 ft³), the mass is
[ 0.25\ \text{ft}^3 \times 263.9\ \frac{\text{lb}}{\text{ft}^3} = 65.975\ \text{lb} ]
This quick calculation informs material cost, transport logistics, and structural analysis.
5. Common Pitfalls to Avoid
| Mistake | Why It Happens | How to Fix It |
|---|---|---|
| Using the wrong mass conversion | Mixing grams with kilograms or pounds incorrectly | Always use 1 g = 0.00220462 lb |
| Ignoring cubic volume conversion | Treating cm³ as ft² or ft³ directly | Multiply cm³ by 0.0000353147 ft³ |
| Rounding too early | Small rounding errors accumulate | Keep at least 5 significant figures until the final step |
| Assuming density is invariant | Temperature or pressure can change density | Verify conditions if precision is critical |
6. Quick Reference Table
| Density (g cm⁻³) | Density (lbs ft⁻³) | Approximate Material |
|---|---|---|
| 1.On the flip side, 0 | 62. Day to day, 9 | Typical Steel Alloy |
| 7. 5 | Aluminum | |
| 4.22 | 263.8 | 486.So 43 |
| 2. 8 | Pure Iron | |
| 8.7 | 168.96 | 560. |
Note: Values are rounded to one decimal place.
7. Frequently Asked Questions
Q1: Can I use a calculator for this conversion?
Yes. Most scientific calculators allow unit conversion. Even so, enter the density in g cm⁻³, then multiply by 62. 4279 to get lbs ft⁻³. Remember to keep the units consistent.
Q2: What if the density is given in kg m⁻³ instead?
1 kg m⁻³ = 0.On the flip side, multiply the kg m⁻³ value by 0. 0624279 lb ft⁻³. 0624279 to get lbs ft⁻³ Not complicated — just consistent..
Q3: Does temperature affect this conversion?
The conversion factor itself is constant. On the flip side, the actual density of a material can change with temperature. For high‑precision work, measure density at the relevant temperature Simple, but easy to overlook..
Q4: Why is the factor 62.4279 and not a round number?
Because it results from the product of two irrational conversion factors (grams to pounds and cubic centimeters to cubic feet). The decimal is exact to the precision of the constants used.
Q5: Is there a mnemonic to remember the conversion?
Think of “62 pounds per foot when 1 g cm⁻³ is the rule.” It’s a simple rhyme that keeps the number in mind.
8. Summary
Converting 4.Here's the thing — 4279, you can reliably translate densities between metric and imperial systems. 22 g cm⁻³** to lbs ft⁻³ is a routine but vital task in many engineering disciplines. By understanding the underlying unit relationships and applying the conversion factor of **62.This skill ensures accurate material calculations, cost estimations, and compliance with industry standards Simple, but easy to overlook. Which is the point..
Use the steps, tables, and FAQ above as a quick reference whenever you encounter density values in either unit system. With practice, the conversion will become second nature, allowing you to focus on the design and analysis that truly matter.
9. Advanced Considerations & Potential Pitfalls
Beyond the straightforward conversion, several nuances demand attention for truly accurate results. Day to day, for critical applications, particularly those involving elevated temperatures or pressures, a more sophisticated approach utilizing density tables or empirical equations is recommended. Deviations from these conditions, particularly temperature and pressure changes, significantly impact material density. Firstly, the provided conversion factor, 62.4279, represents a specific density at standard conditions – approximately 4°C for water. These resources account for the material’s thermal expansion and compressibility.
Secondly, the choice of units – grams per cubic centimeter versus pounds per cubic foot – can introduce subtle errors if not handled meticulously. In practice, maintaining at least five significant figures throughout the conversion, and only rounding to the desired precision at the very end, is crucial for minimizing this effect. As highlighted earlier, rounding too early in the calculation process can accumulate these errors, leading to a noticeable discrepancy in the final result. What's more, be mindful of the underlying assumptions inherent in the conversion; treating cm³ as a direct equivalent to ft³ ignores the dimensional differences, potentially leading to inaccuracies Most people skip this — try not to..
Finally, when dealing with composite materials or mixtures, the overall density is rarely a simple average of the individual component densities. And the arrangement and interaction of the constituent materials play a vital role. Accurate density determination for such systems requires specialized techniques and a thorough understanding of the material’s microstructure And that's really what it comes down to. Worth knowing..
Conclusion:
While the conversion between grams per cubic centimeter and pounds per cubic foot is a fundamental operation in numerous fields, it’s essential to recognize that it’s not a universally precise process. By diligently applying the provided guidelines – utilizing the correct conversion factor, maintaining sufficient precision throughout the calculation, and considering the influence of environmental factors – you can achieve reliable results. That said, for applications demanding the highest levels of accuracy, particularly when dealing with complex materials or extreme conditions, consulting specialized density tables and employing more advanced techniques is critical. Mastering this conversion, coupled with an awareness of its limitations, empowers engineers and scientists to confidently manage the complexities of material properties and ensure the integrity of their designs That alone is useful..